Although evidence-based guidelines for acute bronchiolitis recommend primarily supportive care unnecessary treatments remain well documented. The objective of this study was to analyse a quality improvement initiative to reduce overuse of unnecessary treatments in infants with acute bronchiolitis in primary care settings.
MethodTo determine the number of unnecessary treatments we reviewed the charts corresponding to infants aged <24 months of age diagnosed with acute bronchiolitis in two Primary Care areas during two bronchiolitis seasons [October–March of 2015–2016 (pre-intervention period) and 2016–2017 (post-intervention period)]. Between those seasons we distributed an evidence-based management protocol and developed interactive sessions with on-line data collection and feed-back. Outcomes were the rate of infants receiving salbutamol, steroids or antibiotics.
ResultsTwenty outpatient clinics contributed with 1277 chart reviews (619 in the pre-intervention period and 658 in the post-intervention period). Overall, the use of any medication was reduced from 72.5% (95% CI, 68.8–75.9) to 52.1% (95% CI, 48.3–55.9) (P<.01): salbutamol from 56.0% (95% CI, 52.1–59.9) to 38.3% (95% CI, 34.6–42.0) (P<.01), corticosteroids from 23.7% (95% CI, 20.5–27.2) to 12.9% (95% CI, 10.5–15.7) (P<.01) and antibiotics from 36.1% (95% CI, 32.5–40.0) to 29.6% (95% CI, 26.2–33.2) (P<.05). The number of medications per patient decreased from a mean of 1.81 (SD: 0.86) to 1.62 (SD: 0.81) (P<.01).
ConclusionsWe significantly decreased the use of unnecessary treatments in infants with acute bronchiolitis. This quality improvement initiative may be applied to the settings where the majority of infants with acute bronchiolitis are attended in western countries.
Aunque las guías basadas en la evidencia científica sobre bronquiolitis aguda recomiendan únicamente el tratamiento de soporte, se continúa documentando el uso de tratamientos innecesarios. El objetivo de este estudio fue analizar el impacto de una iniciativa de mejora de la calidad para reducir el sobreuso de tratamientos innecesarios en lactantes con bronquiolitis en atención primaria.
MétodoPara determinar el número de tratamientos innecesarios, fueron revisados los registros correspondientes a niños menores de 2 años de edad diagnosticados de bronquiolitis en dos áreas de atención primaria, durante dos epidemias de bronquiolitis (octubre-marzo de 2015-2016 [periodo preintervención] y de 2016-2017 [periodo postintervención]). Entre estas dos epidemias fue distribuido un protocolo de manejo basado en la evidencia científica y se desarrollaron sesiones interactivas con descripción de datos sobre la utilización de fármacos en los centros implicados. Los resultados fueron la tasa de niños que recibieron salbutamol, corticoides o antibióticos.
ResultadosSe registraron un total de 1.277 episodios de bronquiolitis aguda, atendidos en 20 centros de salud, durante las dos epidemias (619 en el periodo preintervención y 658 en el periodo postintervención). Globalmente, el uso de cualquier medicación se redujo desde un 72,5% (IC 95%: 68,8-75,9) hasta un 52,1% (IC 95%: 48,3-55,9) (p<0,01): salbutamol de un 56,0% (IC 95%: 52,1-59,9) a un 38,3% (IC 95%: 34,6-42,0) (p<0,01), corticoides de un 23,7% (IC 95%: 20,5-27,2) a un 12,9% (IC 95%: 10,5-15,7) (p<0,01) y antibióticos de un 36,1% (IC 95%: 32,5-40,0) a un 29,6% (IC 95%: 26,2-33,2) (p<0,05). El número de medicaciones por paciente disminuyó de una media de 1,81 (DE: 0,86) a 1,62 (DE: 0,81) (p<0,01).
ConclusionesEn el presente estudio, se redujo de forma significativa el uso de tratamientos innecesarios para la bronquiolitis aguda en atención primaria. Esta iniciativa de mejora de la calidad puede ser aplicada en los centros sanitarios donde se atienden la mayoría de los niños con bronquiolitis aguda en los países occidentales.
Acute bronchiolitis (AB) is the most frequent lower respiratory tract infection in children aged less than 2 years. It is a major burden in paediatric emergency care services and is the main reason for hospital admission in infants aged less than 1 year,1,2 with an important health care, economic and social impact.3–5
When it comes to the primary care (PC) level, there is a dearth of data on the incidence of AB and the costs associated with its management. According to data from the Autonomous Community of Valencia, 21% of children in the region have at least one episode of AB secondary to infection by respiratory syncytial virus (RSV) in the first year of life, and 87% of paediatric cases of AB are managed in outpatient care services.6
At present, there is no evidence that drugs or other treatments influence the outcome of AB, and supportive measures are the mainstay of its management.7,8 At the PC level, the management of AB should focus on the appropriate clinical diagnosis and assessment of disease severity and hydration status, the identification of children with risk factors for development of severe disease, and lastly providing support and education to the families of children managed in outpatient services.9
However, despite AB being one of the most frequent diseases in the paediatric age group and of the extensive literature available on its management, including clinical practice guidelines with very restrictive indications for the use of bronchodilators (BDs), corticosteroids or antibiotics, the available evidence recurrently describes a frequent use of unnecessary interventions and drugs, and substantial variability in their prescription.10,11
The aim of our study was to assess the impact of a quality improvement initiative that sought to reduce drug prescription for treatment of AB in two primary care service areas.
Patients and methodsWe conducted a descriptive, community-based pre–post-intervention study with historical controls of 2 consecutive AB epidemic seasons (2015–2016 and 2016–2017) in 2 PC areas in Vizcaya (Basque Country, Spain). We defined the AB epidemic season as ranging from October 1 to March 31. The 2 PC areas comprehend 20 primary care centres (PCCs) with a total of 39 paediatricians in staff currently managing 36,412 children, of who 6371 (17.49%) are aged less than 2 years.
Data collectionTo analyse how AB was managed at the PC level, we collected data from the PCCs included in the integrated health care systems (Organización Sanitaria Integrada [OSI]) of Barakaldo-Sestao and of Ezkerraldea-Enkarterri-Cruces, both in the province of Vizcaya, for 2 bronchiolitis seasons:
- -
Pre-intervention period: 2015–2016 epidemic season (E1).
- -
Post-intervention period: 2016–2017 epidemic season (E2).
We retrieved data from the electronic health records of the Osakidetza (Health Care System of the Basque Country) using the Oracle Business Intelligence (OBI) data mining software. We performed a search for the International Classification of Diseases, Ninth Revision (ICD-9) diagnostic codes for “acute bronchiolitis” (466.1), “acute bronchiolitis due to respiratory syncytial virus” (466.19) and “acute bronchiolitis due to other infectious mechanisms (466.11) in children aged less than 2 years. We only included the first episode with a code corresponding to AB for each patient in each epidemic season. We also collected data on the number of prescriptions of bronchodilators, antibiotics and inhaled and systemic corticosteroids made for home treatment. When it came to prescriptions, we only obtained data for those made through the electronic prescription system that were related to the AB episode.
As control measures, we collected data on the visits related to AB to the paediatric emergency department of the regional referral hospital (Hospital Universitario Cruces) made by children that belonged to the 2 PC areas under study, the severity score received on arrival to the emergency department applying the paediatric version of the Canadian Triage Acuity Scale, and the rate of hospital admission. We retrieved data from the Oracle Business Intelligence (OBI) application of the Osakidetza hospital network. Between the two epidemic seasons, there was a change in the criteria for admission due to AB, eliminating age less than 6 weeks as a sufficient criterion for admission.
Quality improvement initiative- •
Distribution through the mail system of the health care organisation of the protocol recommended for management of AB, which was previously agreed on by the Grupo Interdisciplinar de Emergencias Pediátricas (Interdisciplinary Group on Paediatric Emergency Care [GIDEP]) (available at: http://www.osakidetza.euskadi.eus/r85-pkpubl05/es/contenidos/informacion/gidep_epdt/es_def/index.shtml), a group comprised by PC, hospital paediatric emergency care and emergency response and transport professionals. Health care workers were also reminded of the availability of the protocol through the Intranet of the Osakidetza and other online platforms. In addition, the protocol was distributed in poster format through all the clinics.
- •
Interactive informational sessions were held to be attended by paediatricians. The information was given as PowerPoint presentations and included:
- ∘
Epidemiological data and information on the economic and social impact of RSV and AB.
- ∘
In-house data on the number of episodes of AB managed in the two previous seasons and the use of pharmacological treatment in the different PCCs, focused exclusively on the use of bronchodilators.
- ∘
Reminder of the diagnostic criteria for AB included in the protocol.
- ∘
Key points on current treatment recommendations based on the most recent guidelines of the American Academy of Paediatrics (AAP 2014)7 and the National Institute for Health and Care Excellence (NICE 2015)8 and key points of the recommended protocol. The criteria used to define AB were those proposed by McConnochie12: first acute episode of respiratory distress with wheezing preceded by a cold-like clinical picture of the upper respiratory airway which affects children younger than 2 years of age.
The quality improvement initiative was implemented the month preceding the second epidemic season and was supported at all times by the integrated health care system administration, which facilitated the collection of data, the organisation of the sessions and the dissemination of the protocol.
Statistical analysisWe described qualitative variables as absolute and relative frequencies, and quantitative variables as mean and standard deviation (SD). To analyse the association between qualitative variables, we used the chi squared test. We compared quantitative variables with a normal distribution by means of the Student t test. We performed the statistical analysis with the software SPSS 23.0 (SPSS Inc.; Chicago, IL, USA) for Windows. We defined statistical significance as a P-value of less than .05.
The study was approved by the Clinical Research Ethics Committee of the Basque Country.
ResultsGeneral dataOverall, the incidence of AB in children aged less than 2 years was 9.79% (9.27% in E1 and 10.32% in E2). Combining the two periods, a total of 1277 children—619 in E1 and 658 in E2—received a diagnosis of AB. Of these children, 325 (25.5%) were aged more than 1 year, 609 (47.7%) were aged 6 to 12 months and 343 (26.8%) were aged less than 6 months. A total of 792 children (64.5%) received drug prescriptions: for bronchodilators in 599 (48.8%), oral corticosteroids in 232 (18.9%), antibiotics in 419 (34.1%) and inhaled corticosteroids in 113 (9.2%). Drugs were prescribed to 263 of the children aged more than 1 year (80.9%; 95% CI, 76–84) versus 338 of the infants aged 6 to 12 months (55.5%; 95% CI, 51–59) and 181 of the infants aged less than 6 moths (52.7%; 95% CI, 47–57) (P<.01). The mean number of different drugs prescribed per patient was 1.72 (SD, 0.84), and more than 1 drug was prescribed in 402 children (50.7%). Children aged more than 1 year received a mean of 1.83 prescriptions (SD, 0.82), compared to 1.67 (SD, 0.84) in infants aged less than 12 months (P<.01). At least 2 drugs were prescribed in a total of 158 children aged more than 1 year (60.5%; 95% CI, 55–65) compared to 247 infants aged less than 1 year (46.3%; 95% CI, 42–49) (P<.01). In the subset of infants aged less than 6 months, 77 (42.5%; 95% CI, 35–49) received more than 1 drug compared to 170 (50.3%; 95% CI, 44–55) in the subset of infants aged 6 to 12 months (P<.01).
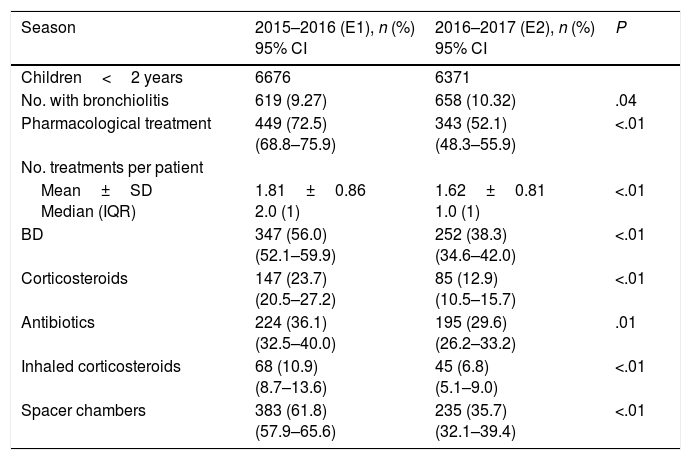
Comparison of the two epidemic seasonsDuring E1, pharmacological treatment was prescribed to 449 (72.5%) children compared to 343 (52.1%) in E2 (P<.01). When it came to the drugs prescribed, bronchodilators were prescribed to 347 patients (56%) in E1 compared to 252 patients (38.3%) in E2 (P<.01). The mean number of drugs prescribed by patient was 1.81 (SD, 0.86) in E1 versus 1.62 (SD, 0.81) in E2 (P<.01). Table 1 shows the other drugs and inhaler devices prescribed in the two epidemic seasons.
Percentage of patients that received drug prescriptions overall, mean number of prescribed drugs per patient, and percentage of patients that received drug prescriptions specifically for bronchodilators, oral corticosteroids, antibiotics, inhaled corticosteroids and spacer chambers, in the 2 epidemic seasons.
| Season | 2015–2016 (E1), n (%) 95% CI | 2016–2017 (E2), n (%) 95% CI | P |
|---|---|---|---|
| Children<2 years | 6676 | 6371 | |
| No. with bronchiolitis | 619 (9.27) | 658 (10.32) | .04 |
| Pharmacological treatment | 449 (72.5) (68.8–75.9) | 343 (52.1) (48.3–55.9) | <.01 |
| No. treatments per patient | |||
| Mean±SD Median (IQR) | 1.81±0.86 2.0 (1) | 1.62±0.81 1.0 (1) | <.01 |
| BD | 347 (56.0) (52.1–59.9) | 252 (38.3) (34.6–42.0) | <.01 |
| Corticosteroids | 147 (23.7) (20.5–27.2) | 85 (12.9) (10.5–15.7) | <.01 |
| Antibiotics | 224 (36.1) (32.5–40.0) | 195 (29.6) (26.2–33.2) | .01 |
| Inhaled corticosteroids | 68 (10.9) (8.7–13.6) | 45 (6.8) (5.1–9.0) | <.01 |
| Spacer chambers | 383 (61.8) (57.9–65.6) | 235 (35.7) (32.1–39.4) | <.01 |
Of the children to whom pharmacological treatment was prescribed, 124 (56%; 95% CI, 50–63) received prescriptions for more than 1 drug in E1, compared to 66 (42%; 95% CI, 34–49) in E2 (P<.01). This decline mainly corresponded to patients aged less than 1 year, as the proportion dropped from 53% (95% CI, 45–62) in E1 to 36% (95% CI, 27–46) in E2 (P=.03).
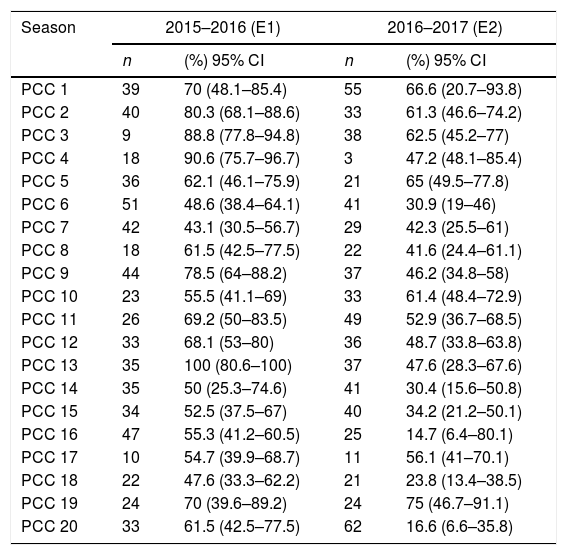
Variability between primary care centresTable 2 shows the number of patients that received a diagnosis of AB and the percentage of patients given a prescription for pharmacological treatment in each PCC and in each season under study. In both seasons, the percentage of patients that received a prescription for pharmacological treatment varied between PCCs and ranged between 30% and 100% and, for the specific case of bronchodilators, between 15% and 90%. In the group of patients that received prescriptions for pharmacological treatment overall, the mean number of drugs prescribed per patient in individual PCCs ranged from a minimum of 1.35 (SD, 0.55) to a maximum of 2.20 (SD, 1.01).
Number of patients with a diagnosis of AB (n) and proportion of patients that received drug prescriptions overall (% and 95% CI), in the participating primary care centres (PCCs) for each of the 2 epidemic seasons.
| Season | 2015–2016 (E1) | 2016–2017 (E2) | ||
|---|---|---|---|---|
| n | (%) 95% CI | n | (%) 95% CI | |
| PCC 1 | 39 | 70 (48.1–85.4) | 55 | 66.6 (20.7–93.8) |
| PCC 2 | 40 | 80.3 (68.1–88.6) | 33 | 61.3 (46.6–74.2) |
| PCC 3 | 9 | 88.8 (77.8–94.8) | 38 | 62.5 (45.2–77) |
| PCC 4 | 18 | 90.6 (75.7–96.7) | 3 | 47.2 (48.1–85.4) |
| PCC 5 | 36 | 62.1 (46.1–75.9) | 21 | 65 (49.5–77.8) |
| PCC 6 | 51 | 48.6 (38.4–64.1) | 41 | 30.9 (19–46) |
| PCC 7 | 42 | 43.1 (30.5–56.7) | 29 | 42.3 (25.5–61) |
| PCC 8 | 18 | 61.5 (42.5–77.5) | 22 | 41.6 (24.4–61.1) |
| PCC 9 | 44 | 78.5 (64–88.2) | 37 | 46.2 (34.8–58) |
| PCC 10 | 23 | 55.5 (41.1–69) | 33 | 61.4 (48.4–72.9) |
| PCC 11 | 26 | 69.2 (50–83.5) | 49 | 52.9 (36.7–68.5) |
| PCC 12 | 33 | 68.1 (53–80) | 36 | 48.7 (33.8–63.8) |
| PCC 13 | 35 | 100 (80.6–100) | 37 | 47.6 (28.3–67.6) |
| PCC 14 | 35 | 50 (25.3–74.6) | 41 | 30.4 (15.6–50.8) |
| PCC 15 | 34 | 52.5 (37.5–67) | 40 | 34.2 (21.2–50.1) |
| PCC 16 | 47 | 55.3 (41.2–60.5) | 25 | 14.7 (6.4–80.1) |
| PCC 17 | 10 | 54.7 (39.9–68.7) | 11 | 56.1 (41–70.1) |
| PCC 18 | 22 | 47.6 (33.3–62.2) | 21 | 23.8 (13.4–38.5) |
| PCC 19 | 24 | 70 (39.6–89.2) | 24 | 75 (46.7–91.1) |
| PCC 20 | 33 | 61.5 (42.5–77.5) | 62 | 16.6 (6.6–35.8) |
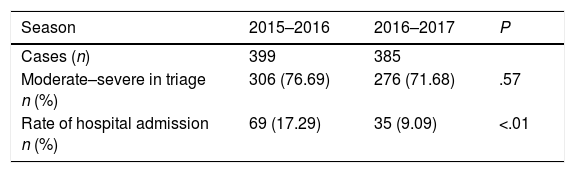
Table 3 presents the data on the frequency of the AB-related visits to the emergency department of the referral hospital by children belonging to the 2 PC areas under study, in addition to the severity level at triage and rate of hospital admission, for each of the epidemic seasons. A total of 69 children (17.3%) were admitted during E1 compared to 35 (9.1%) during E2 (P<.01).
Visits related to AB to the emergency department of the referral hospital, disease severity and rate of hospital admission in children belonging to the 2 primary care service areas under study for each of the 2 epidemic seasons.
| Season | 2015–2016 | 2016–2017 | P |
|---|---|---|---|
| Cases (n) | 399 | 385 | |
| Moderate–severe in triage n (%) | 306 (76.69) | 276 (71.68) | .57 |
| Rate of hospital admission n (%) | 69 (17.29) | 35 (9.09) | <.01 |
Our study demonstrated that the implementation of a quality improvement intervention at the level of a primary care service area can succeed in reducing the use of pharmacological treatment in the management of AB. However, it also showed that a very high percentage of infants and toddlers with AB continued to be treated with bronchodilators, corticosteroids and antibiotics despite evidence that they are not useful in the management of this disease.13,14 Furthermore, there was considerable variability between clinicians in the prescription of the different drugs, even in a geographical area as small as the PC areas under study.
Clinical practice guidelines are excellent vehicles to inform about good practices and the current scientific evidence on the management of a disease, in this case AB, but their development and dissemination in the scientific literature have proven insufficient to drive change in the clinical practice in many professionals.15,16 Previous studies conducted in hospital settings have demonstrated the usefulness of implementing quality improvement measures at a local level or with collaboration between different settings to improve the management of AB.17–19 Many of the interventions designed to improve the management of AB combine the development of guidelines or local protocols with strategies for their implementation and the education of health care professionals. A study conducted by Ralston et al. to assess a collaborative multicentre strategy18 found a significant decrease in the use of bronchodilators and corticosteroids after the dissemination of the AAP guidelines and education through interactive webinars. Similarly, multifaceted educational bundles, such as the one described by Murch et al.,19 can significantly improve adherence to guidelines. However, no studies to date have been published on similar strategies at the primary care level. Our study shows that approaches similar to those used in hospital settings can achieve a change in clinical practice, in this case, a reduction of 20% in the use of drugs for treatment of AB in PC. The key elements were, on one hand, the dissemination and implementation of a protocol underscoring the most important aspects in the management of children with AB at the PC level, with a restrictive approach regarding the use of bronchodilators in adherence with current recommendations, and on the other hand, and more importantly, the feedback provided to health professionals through the presentation of data on their own clinical practice. This last part is crucial, as there are few published case series of patients managed at the PC level,6,11,20 and professionals need to have access to epidemiological and management data for this care setting.
Multiple studies have called attention to the frequent use of unnecessary interventions in the management of AB.10,21,22 In our study, the excessive use of medication continued to be a problem, since at its conclusion, 52% of children with AB in the PC service area under study continued to receive pharmacological treatment, with a pattern of use that was similar to the patterns reported in the PC literature,11 with the frequent use of combinations of more than 1 drug in a single child (52.3%). It is obvious that there is still ample room for improvement until we reach the awareness that “less is more in bronchiolitis”,23 but this is all the more worrisome at the primary care level, where such a substantial use of drugs can hardly be justified. An interesting finding in our study was that, overall, children aged more than 1 year received a greater number of drugs per patient, 1.84 compared to 1.69 in infants, and that a higher proportion of those aged more than 1 year received more than 1 drug—60.27% compared to 47.5% of infants. This difference is probably due in part to the diagnostic criteria used by clinicians. The more restrictive use of drugs in infants may reflect a better adherence to diagnostic criteria and easier coding and electronic charting in this age group. Children with a previous diagnosis of AB in the first year of life can receive this diagnosis one more time, but are to be managed as children aged more than 2 years with recurrent wheezing episodes, which nowadays would be included under the label of asthma in infants and young children.
Another salient finding in our study was the considerable variability between professionals in the prescription of drugs for treatment of AB. We ought to highlight that while the variability in the prescription of drugs is well known and has been repeatedly described in the literature,10,22 it is not only present between different hospitals within a single country24–26 or a single autonomous community,11 but also, as our study evinced, between PCCs in the same health care system. Furthermore, we found that the PCCs with the highest use of drugs per patient at the outset continued to show this trend after the intervention, which is indicative of the challenges involved in changing long-standing clinical practices. Since the main reason for the excessive use of drugs is not insufficient knowledge of the guidelines, but their modest effectiveness in changing the habitual practices of clinicians, our aim should be not only to update them, but to achieve the maximum impact by developing strategies for their implementation and promoting adherence.22,27 It is key that health care systems become involved in this regard and provide health professionals feedback on their clinical activity, giving them tools for its management, such as in-house indicators and quality standards.
Lastly, we need to improve the knowledge of the general population about AB. In a recent survey,28 19% of clinicians attributed the excessive use of drugs for treatment of AB to the demands made by the family. It is likely that a better understanding of AB, its presentation and its management on the part of parents would facilitate care delivery and reassure both the family and the clinician in charge of its management.
There are limitations to our study. First of all, it only included two PC service areas, so the characteristics of other health care systems could lead to different outcomes of the same intervention. Second, we obtained data retrospectively, which may have resulted in incomplete, heterogeneous or unreliable data. However, we retrieved the data from an electronic system where clinical interventions were recorded at the time they were performed and that was not related to the quality improvement initiative. Third, since this was a community-based pre–post-intervention study, the decline in drug prescription could be due to factors other than the implemented intervention. We do not believe that a greater severity of disease in the second epidemic season was one such factor, as the severity at triage of children managed in the emergency department was the same in both seasons. Fourth, since we only obtained data on drug prescriptions for at-home use and did not investigate the potential administration of bronchodilators at the time of the PC visit, we may have underestimated pharmacological treatment overall. We believe that this potential underestimation would not affect the primary outcome of this intervention, which is the decrease in prescription. Lastly, we did not review patient health records to assess whether clinicians used the McConnochie criteria for diagnosis of AB, as recommended by the established protocol, so that differences between professionals in the applied diagnostic criteria led to overestimation of the incidence of AB. However, the incidence found in our sample was similar to those reported in other studies.
ConclusionsQuality improvement measures implemented at the local level to reduce the use of drugs in the management of AB seem to be effective. Nevertheless, there is still frequent use of pharmacological treatment and great variability between clinicians in the management of the disease, which evinces the need to maintain the current quality improvement interventions and launch new ones. The intervention implemented in this study could be used in other PCCs, where most children with AB are managed in Western countries.
Conflicts of interestThe authors have no conflicts of interest to declare.
Please cite this article as: Montejo Fernández M, Benito Manrique I, Montiel Eguía A, Benito Fernández J. Una iniciativa para reducir el uso de medicación innecesaria en lactantes con bronquiolitis en atención primaria. An Pediatr (Barc). 2019;90:19–25.