At present there is a degree of uncertainty regarding when, how and in what form gluten should be introduced into the infant diet. For years the recommendations of the ESPGHAN Committee on Nutrition have prevailed, which include avoiding early introduction, before 4 months, and late, after 7 months, and gradually introducing gluten into the diet while the infant is being breastfed, with the aim of reducing the risk of celiac disease, diabetes and gluten allergy. However, 2 independent studies published in The New England Journal of Medicine in October 2014 reached the conclusion that the age of introduction of gluten does not modify the risk of developing celiac disease, and that breastfeeding at any age does not confer protection against celiac disease development.
On the other hand, according to available scientific evidence, the introduction of foods other than breast milk or formula into the infant diet is generally recommended around 6 months of age, since the introduction before 4 months could be associated with an increased risk of food allergy and autoimmune diseases, and delaying it beyond 7 months would not have a protective effect.
In this context, a group of experts has considered it appropriate to produce a consensus document based on the current scientific evidence and present general recommendations for daily clinical practice on the introduction of gluten into the diet.
En el momento actual existe una situación de indefinición con respecto a cuándo, cómo y de qué forma debe introducirse el gluten en la dieta del lactante. Durante años ha prevalecido la recomendación del Comité de Nutrición de la ESPGHAN de evitar tanto la introducción precoz, antes de los 4 meses, como la tardía, después de los 7 meses, y de introducir el gluten gradualmente mientras el lactante recibe leche materna; se pretendía con ello reducir el riesgo de enfermedad celiaca, diabetes y alergia al gluten. Sin embargo, 2 estudios independientes publicados en octubre de 2014 en The New England Journal of Medicine llegan a la conclusión de que la edad de introducción del gluten no modifica el riesgo de desarrollar la enfermedad celiaca y que la lactancia materna a cualquier edad tampoco confiere protección.
Por otra parte, según la evidencia científica disponible, en general, se recomienda la introducción de otros alimentos en la dieta distintos de la leche materna o de fórmula alrededor de los 6 meses de edad, ya que la introducción antes de los 4 meses se asociaría a un riesgo aumentado de enfermedades autoinmunes y alergia alimentaria, y retrasarla más allá de los 7 meses no tendría efecto protector.
En este contexto, un grupo de expertos ha considerado pertinente elaborar un documento de consenso basado en las evidencias científicas actuales y establecer unas recomendaciones generales para la introducción del gluten en la práctica clínica diaria.
For more than 20 years the ESPGHAN has made, through its Nutrition Committee (CN-ESPGHAN), recommendations related to the age of gluten introduction into an infant's diet1,2 with the purpose of preventing the development of coeliac disease (CD) and reducing the risk of diabetes mellitus type 1 (DM 1) and wheat allergy.2,3
However, recent studies of reliable evidence have demonstrated that the age of gluten introduction does not influence the development of CD in the population with genetic risk,4,5 calling into question the current recommendations of the ESPGHAN,2 the European Food Safety Authority6 and the most recent recommendations of the American Academy of Paediatrics. The latter recommends the introduction of complementary feeding (CF) around 6 months, without making special mention of gluten.7
Due to the current confusion, a group of experts in CD and paediatric nutrition have prepared a consensus document based on the current scientific evidence, establishing some recommendations for daily clinical practice.
General aspects of the introduction of complementary feedingThe recommendations of the ESPGHAN, the European Food Safety Authority and the American Academy of Paediatrics regarding the introduction of CF take into consideration, apart from the nutritional needs, the possibility of influencing the development of certain diseases.2,6,7
Allergy preventionThere are no scientific data supporting different recommendations for the introduction of CF based on an infant's risk of suffering from allergies (first degree relative with proven history of allergy).8–11
The introduction of CF after 6 months has no protective effect on the appearance of food allergy.12 On the contrary, if it is introduced before 4 months it is associated with an increased risk of developing atopic dermatitis.
Neither has it been proven that there is an increasing risk of developing food allergies when the most potentially allergising foods are included in the diet after 4 months.10
Regardless of the time of introduction, after incorporating a new food, it seems prudent to recommend a regular exposure (for instance, several times a week) to maintain oral tolerance. It has also been observed that the inclusion of a wider diversity of food during the first year is associated with a decreased risk of asthma, food allergies or food sensitisation.13
Development of obesityA high protein intake, especially from animal origin and during the first 2 years of life, is associated with an increasing risk of obesity later.14,15 There is no evidence on when to introduce CF to diminish the risk of obesity.16
Development of diabetes mellitus type 1It has been found that there is an increased risk of developing DM 1 when the CF is introduced before 4 months or after 6 months.17 However, no specific dietary factor has been shown to have a higher risk.18
To conclude, according to scientific evidence, the introduction of CF before 4 months is associated with an increased risk of disease, whereas the introduction after 7 months has no protective effect; therefore, the advice to introduce CF around 6 months prevails.
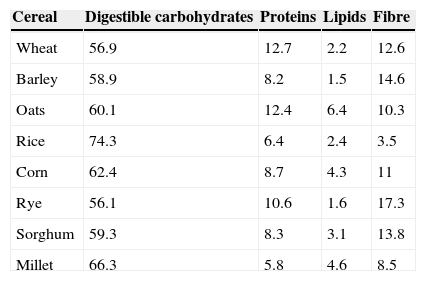
Nutritional value of cerealsCereals, one of the bases of the human diet, represent the main source of carbohydrates (CH) and fibre in diet, the most common being: wheat, rice, corn, rye, barley, oats, millet and sorghum.19 They contain around 70–78% of CH, 6–13% of proteins and 1–6% of fats (Table 1).19,20
Composition of cereals.
| Cereal | Digestible carbohydrates | Proteins | Lipids | Fibre |
|---|---|---|---|---|
| Wheat | 56.9 | 12.7 | 2.2 | 12.6 |
| Barley | 58.9 | 8.2 | 1.5 | 14.6 |
| Oats | 60.1 | 12.4 | 6.4 | 10.3 |
| Rice | 74.3 | 6.4 | 2.4 | 3.5 |
| Corn | 62.4 | 8.7 | 4.3 | 11 |
| Rye | 56.1 | 10.6 | 1.6 | 17.3 |
| Sorghum | 59.3 | 8.3 | 3.1 | 13.8 |
| Millet | 66.3 | 5.8 | 4.6 | 8.5 |
Approximate composition of cereals (g/100g of edible portion).
Starch is the main CH of cereals, which also contain other non-digestible polysaccharides, components of the dietary fibre. CH must represent between 45 and 65% of the total caloric value of an infant's diet (Table 2).21,22
Although cereals contain all the amino acids, they show relative deficiencies of some of them, which gives them a low biological value, and they must be supplemented with other proteins. The protein content varies according to the type of cereal, being higher in wheat and oats and lower in rice and corn.19
Cereals are an important source of vitamins from group B (niacin and thiamine),23 of small amounts of trace elements (iron and selenium) and minerals (phosphorus, potassium, magnesium and calcium).
Gluten in cerealsGluten is, due to its cohesive and viscoelastic nature, especially relevant in bread-making processes.24 It contains thousands of proteins rich in glutamine and proline, called prolamines for that reason25; these are just partially digested by human proteases, a relevant aspect in the pathogenesis of the CD.
The prolamines of wheat (gliadins), rye (secalins) and barley (hordeins) are related to CD, while the role of oat prolamines (avenins) is controversial. Nevertheless, generally wheat, barley, rye and oats are considered cereals with gluten. Gluten represents 80% of proteins. The proportion of prolamines varies; it is very high in wheat compared to in other cereals.
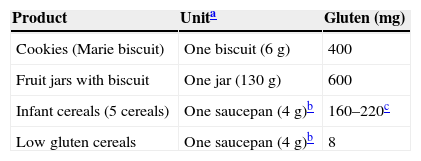
Usually, cereals are the first food added after milk to an infant's diet.2 The gluten content of the most common food used at the beginning of CF is shown in Table 3.
There are no nutritional reasons that justify the consumption of a particular cereal. Generally, it is recommended to start the CF with gluten-free cereals because they have lower antigenic capacity. The consumption of a particular cereal is determined by cultural considerations: wheat is preferentially consumed in Europe and western countries, corn in America and rice in eastern countries.
Recommendations of the Nutrition Committee of the European Society for Paediatric Gastroenterology, Hepatology and NutritionIn 1982, the CN-ESPGHAN,1 following previous recommendations from 1969, issued the following general warning: “Gluten should not be introduced to infants before 4 months, and it is highly recommended to postpone it until 6 months.” These recommendations were based on the fact that sensitisation to gluten could possibly be more easily induced in smaller infants, particularly in those artificially fed.
These regulations were in force until 2008, when the CN-ESPGHAN2 updated them based on:
- •
A systematic review and meta-analysis that showed that the risk to develop CD was significantly reduced in those infants who were breastfeeding (BF) at the time of introduction of gluten compared to those who did not breastfeed, and that longer duration of BF was associated with a lower risk of CD. However, it was not proven that BF gave permanent protection against CD or just delayed the age of appearance.26
- •
The so-called “Swedish epidemic” of CD: this sudden increase in the incidence of CD in children <2 years was detected between 1984 and 1996, matching changes in the introduction of gluten from 6 months and an abrupt introduction of large amounts of wheat. Therefore, at 6–8 months of age, wheat consumption increased from 10 to 26g/day (equivalent to an increase from 0.9 to 2.7g of gluten/day). At the end of the 1990s, after recommending again the introduction of gluten at the fourth month and reducing ≥30% the wheat consumption, a significant fall in the number of CD cases in this age group was observed.27
- •
A prospective study (Norris et al.) that showed, in children with a first degree relative with DM 1 or bearing a high risk HLA (DR3 or DR4), that the introduction of gluten very early (<3 months) or late (>7 months), increased the risk of CD.28
Based on these studies, the CN-ESPGHAN advised: “It is prudent to avoid the early (<4 months) and late (>7 months) introduction of gluten and gradually introduce it while an infant receives BF, since the risk of CD, DM 1 and gluten allergy can be reduced in that way.”2 This recommendation was confirmed by the European Food Safety Authority6 and a systematic review of infant feeding and CD prevention,3 although more studies were necessary to clarify unresolved issues. In fact, the Scientific Advisory Committee on Nutrition (SACN) and the Committee on Toxicity (COT) of the United Kingdom29 specified that there was not enough scientific evidence to make specific recommendations about the introduction of gluten into infant feeding. Indeed, recent Swedish studies show that, although the previous regulations on the introduction of gluten had not changed, the number of CD cases increased again by the end of the 1990s. From that moment on, a cumulative incidence similar to the one in the 1980s, years of the epidemic, was observed.30,31
To conclude, the current recommendations of the CN-ESPGHAN related to when -from 4 to 7 months-, how -progressive increase from small amounts- and in which circumstances -while the infant receives BF- should gluten be introduced are apparently not enough for the primary prevention of CD.
Introduction of gluten and risk of coeliac diseaseThe hypothesis of a window between 4 and 7 months during which the introduction of small amounts of gluten while the child still receives BF could protect from the development of CD in children at risk3,27,30–33has been recently addressed by 2 studies published in The New England Journal of Medicine.4,5 In addition, a publication included in the same edition emphasises the relevance of the reported findings.34
In the prospective study PreventCD (www.preventcd.com), babies with CD risk were chosen (first degree relative with CD and at least one risk HLA: DQ2 and/or DQ8).4 These were randomly (blindly) separated into 2 groups, receiving from 16 weeks of age 200mg of gluten (containing 100mg of immunogenic gluten), daily for 8 weeks, group 1, or a placebo (2g of lactose), group 2. BF was recommended throughout this period. From 24 weeks on, the amount of gluten was increased using normal products in child feeding (cereals or biscuits), following a pre-established protocol: between 6 and 7 months, 250mg of gluten per day; between 7 and 8 months, 500mg; between 8 and 9 months, 1000mg; between 9 and 10 months, 1500mg; and free consumption from 11 months onwards. Nine hundred and ninety-four children complied with this protocol, which included this clinical follow-up: indicative signs or symptoms of CD,35 detection of anti-transglutaminase antibodies, growth and diet control (BF and consumption of gluten). When all the children were 3 years old, the codes were opened and the 2 groups were separately analysed. The CD frequency in the cohort at 3 years (cumulative incidence) was 5.2%. Differences between group 1 and group 2 were not observed. No relationship was found between the development of CD and duration of BF (exclusive or mixed) or the continuation of BF during the introduction of gluten. However, it was observed that DQ2 homozygous infants (DR3-DQ2/DR3-DQ2 or DR3-DQ2/DR7-DQ2) had a significantly higher risk to develop CD before 3 years than infants with lower risk haplotypes.
In addition, at 3 years the frequency of CD in girls doubled the frequency in boys: 7.2% vs 3.4%. What's more, a higher, statistically significant, cumulative incidence of CD was detected in girls who were introduced to gluten at 16 weeks compared to those who were introduced at 24 weeks. This difference was not observed in boys.
The authors conclude that “the introduction of small amounts of gluten between 16 and 24 weeks did not reduce the risk of CD”.
The multicentre study CELIPREV is published in the same edition of the New England Journal of Medicine. In it, newborns with a risk of CD (at least one first degree relative with CD) were selected and randomised: one group started the intake of gluten at 6 months (pasta, semolina and biscuits), group 1, and the other one, from the age of 12 months, group 2.5 The amount of gluten consumed by infants was free. During the first year, information about intestinal infections and diet was gathered (daily content of gluten) and HLA genotyping was performed at 15 months (excluding DQ2-DQ8 negative). Antigliadin and antitransglutaminase antibodies at 2, 3, 5, 8 and 10 years in the 553 boys finally included. At 2 years, boys from group 1 had developed CD in a significantly higher percentage (12% vs. 5%) than those from group 2. At 5 and 10 years the differences between the 2 groups were not statistically significant. At 10 years, 25.8% of the homozygote boys DQ2/DQ2 had developed CD vs. 15.8% of those who had lower risk phenotypes. BF was not associated with the development of CD. Although the authors state that even though delaying the beginning of CD could have some benefit, there is no evidence for supporting this premise.
The authors conclude that “neither the late introduction of gluten nor BF modified the risk of CD between boys at risk, even though the late introduction of gluten was associated with a delay in the appearance of the disease”.
Conclusions of both studies- •
The age at gluten introduction does not modify the risk of developing CD; therefore, neither of the 2 studies support the hypothesis of an age window that favours developing tolerance to gluten. Introduction from age 12 months could delay the beginning of CD.
- •
The genetic phenotype is the most determinant risk factor for developing CD at an early age, especially for homozygotes DQ2/DQ2.
- •
BF does not protect against the development of CD.
The introduction of CF from 4 months does not relate to the development of food allergy or DM 1, although if it is hyperproteic it could favour the development of obesity.
The introduction of cereals is justified due to their nutritional properties, based on the selection of the cereal according to the population's habits.
There is no evidence that different or specific recommendations are necessary for the introduction of CF in infants at risk of allergy or CD.
The duration of exclusive BF and the moment of the introduction of CF are not related to the appearance of CD or food allergy.
The introduction of small amounts of gluten at an early age does not reduce the risk of CD in boys at genetic risk. Delaying the introduction of gluten does not modify the risk of CD in infants with genetic predisposition either, even though the late introduction could delay the age of appearance of the disease.
Due to the lack of recent studies, the highest risk of autoimmunity is associated with the introduction of gluten before 4 months in populations with a genetic predisposition. Moreover, the data of the PreventCD study in girls at high genetic risk of CD recommend caution in the introduction of gluten to this sub-population before 6 months.
To date, there is no recommendation by scientific societies on the particular amount of gluten to introduce. In Sweden, in the years before the epidemic, infants consumed 0.9g of gluten/day; during the epidemic, 2.5–2.7g/day and about 2g/day in the years after it. In countries with lower prevalence of CD such as Denmark, at that period (1987), consumption was about 0.2g/day at 6–8 months and 1.8g at 9–12 months. Nevertheless, these data are estimates based on sales of infant cereals in the corresponding years and the nutritional recommendations of that time; they are not based on dietary surveys of real consumption. Also, the PreventCD study shows that consumption of only 200mg of gluten can induce an immunological response.
Neither has it been observed that the introduction of gluten while BF continues to protect from the development of CD.
A wider variety of food in the diversification during the first year of life can have a protective effect on the development of allergic diseases.
To conclude, although the recently published studies do not support the current recommendations of the CN-ESPGHAN regarding the introduction of gluten in an infant's diet, they do not invalidate them either. Neither do they give sufficient evidence to establish new recommendations. We do not know the appropriate age for the introduction of gluten into an infant's diet nor the appropriate way to administer it in the general population and risk population.
Recommendations of the group of expertsAlthough there is not enough scientific evidence, based on the current knowledge (studies of nutrients’ requirements and studies on impact for early feeding and disease prevention) the group of experts reached a consensus on the following aspects, for the general population and at-risk population:
- 1.
BF is always advisable regardless of its effect on the development of CD.
- 2.
It is recommended to preferably introduce CF while the infant is still being breastfed.
- 3.
The introduction of gluten before 4 months is not recommended.
- 4.
It is recommended to introduce gluten around the age of 6 months:
- a.
Introduction between 5 and 6 months seems a reasonable option.
- b.
If for any reason the complementary feeding is started early, it could eventually be considered to introduce gluten after 4 months, even though at that age other cereals, such as corn or rice, unrelated to CD, could be used.
- c.
In case of delaying the introduction of CF, the introduction of gluten could be delayed without implying an additional risk to the patient regarding the possible development of CD.
- 5.
Introduce gluten in small amounts: start with 1–2 small spoons of cereal or one biscuit per day, or equivalent amounts of gluten (Table 3).
- 6.
Once it is introduced, increase consumption gradually according to nutritional needs (caloric intake) and infant's tolerance.
- a.
This work has not been funded.
Conflict of interestThere is no conflict of interests.
Please cite this article as: Ribes Koninckx C, Dalmau Serra J, Moreno Villares JM, Diaz Martín JJ, Castillejo de Villasante G, Polanco Allue I. La introducción del gluten en la dieta del lactante. Recomendaciones de un grupo de expertos. An Pediatr (Barc). 2015;83:355.e1–355.e7.