The objective of the study was to analyse the correlation between extracorporeal life support (ECLS) and aortic cross-clamp times and optic nerve sheath diameter (ONSD).
Patients and methodsStudy in a cohort of patients aged 0–15 years that underwent ECLS for cardiac surgery after obtention of signed informed consent. We calculated a sample size of 23 participants. First, we obtained 3 vertical and 3 horizontal measurements of the ONSD for each eye and calculated the mean of both eyes for each measurement to be used in the analysis. The measurements were made at admission and at 6 and 24 h post surgery. We retrieved the ECLS time and the aortic cross-clamp time were from the operative report.
ResultsWe analysed data for 23 participants, 52.2% female, with a median age of 14 months. The median ECLS time was 60 min; the median aortic cross-clamp time was 32 min. The median baseline ONSD was 3.1 mm. ONSD values had increased a median of 0.015 mm at 6 h post surgery (P = .03). We found a positive correlation between ECLS time and ONSD values (r = 0.476, P < 0,05). The ONSD values returned to baseline by 24 h post surgery. None of the patients developed signs or symptoms of increased intracranial pressure.
ConclusionOur study found a correlation between ECLS time and ONSD at 24 h post surgery. We found variations in the ONSD even in patients without signs or symptoms of increased ICP. Further research is required to identify the factors related to these variations.
El objetivo de este estudio es establecer la correlación entre los tiempos de circulación extracorpórea (CEC) y pinzamiento aórtico y el diámetro de la vaina del nervio óptico (DVNO).
Pacientes y métodosSe estudió una cohorte de pacientes con edad de 0 a 15 años, sometidos a cirugía cardiaca con CEC. Se calculó un tamaño muestral de 23 participantes. Primero se obtuvo el promedio de tres mediciones verticales y tres horizontales del DVNO. Después, se utilizó para los análisis la media de los valores de ambos ojos. Las mediciones ocurrieron al ingreso y a las 6 y 24 horas poscirugía. Los tiempos de CEC y pinzamiento aórtico se obtuvieron del informe quirúrgico.
ResultadosSe analizaron datos de 23 participantes, el 52,2% mujeres, con edad mediana de 14 meses. La mediana del tiempo de CEC fue 60 minutos; la mediana de tiempo de pinzamiento aórtico fue 32 minutos. La mediana del DVNO basal fue 3,1 mm. Los valores de DVNO aumentaron por una mediana de 0,015 mm a las 6 horas poscirugía (p = 0,03). Encontramos una correlación positiva entre el tiempo de CEC y los valores de DVNO (r = 0,476; p < 0,05). Los valores de DVNO regresaron al valor basal a las 24 horas poscirugía. Ningún paciente desarrolló signos o síntomas de incremento de la presión intracraneal.
ConclusiónEl estudio encontró una correlación entre el tiempo de CEC y el DVNO medido 24 horas postcirugía. Hubo variaciones del DVNO incluso en pacientes sin signos o síntomas de incremento en la PIC. Se requiere más investigación para identificar los factores relacionados con estas variaciones.
According to the international report of the Pediatric Extracorporeal Life Support Organization Registry, the number of children that receive extracorporeal life support (ECLS) is increasing, especially for cardiac indications.1 It is also well known that a high proportion of children that undergo ECLS for paediatric cardiac surgery suffer complications, such as need of renal support, bleeding, sepsis, sternal reopening, and cardiac arrest.2 While severe neurological complications are relatively rare (incidence of stroke, 5%–14%, and incidence of brain death, 2%),1 evidence has recently emerged of the importance of more subtle neurologic findings during the long-term follow-up.3 The incidence of post-ECLS cerebral dysfunction, which can lead to permanent cognitive and motor disability and in its earliest stages manifests with brain oedema and elevated intracranial pressure (ICP), can be as high as 70%.4
Due to the limited specificity of the physical examination in neonates and infants, brain oedema in the early postoperative period of cardiac surgery may be clinically silent. Thus, the identification and diagnosis of post-ECLS cerebral dysfunction and more severe neurological injuries that manifest with early brain oedema currently rely on ancillary tests.5 Imaging techniques such as magnetic resonance imaging (MRI) or computed tomography (CT) have the disadvantage of requiring transport of a critically ill patient to the imaging area. On the other hand, invasive monitoring of ICP (by means of an intraventricular catheter or intraparenchymal microsensor system) is associated intrinsically with an increase in morbidity and mortality. Therefore, non-invasive alternatives such as bedside optic nerve ultrasound are preferable options for the early detection of the neurological complications of ECLS.6
The measurement of the optic nerve sheath diameter (ONSD) in the point-of-care ultrasound examination for assessment of critically ill patients, and especially for ICP monitoring, is becoming an increasingly widespread practice. Although the evidence on the usefulness of the ONSD measurement seems to be restricted to acute elevations of ICP, this parameter has exhibited a good correlation with the ICP.
Previous studies have reported that longer ECLS times and aortic cross-clamp times are associated with a higher frequency of detection of cerebral oedema by CT.4,7 However, to date, there are no data on the association of these time markers and the ONSD measured by ultrasound. Therefore, the objective of our study was to analyse the association of the ECLS time and the aortic cross-clamp time with the ONSD in children that receive ECLS.
Material and methodsApplying the formula for calculation of the sample size for correlation studies described by Hulley et al.,8 we estimated that we needed a sample of 23 patients for an alpha level of 0.50, a beta level of 0.20 (2-tailed test) and an expected correlation coefficient of 0.56.
The study protocol was approved by the institutional review board of bioethics (reference no: CEI/59/19). We obtained signed informed consent from parents before enrolling each of the participants.
Between April and October 2019, we consecutively enrolled all patients aged 0 tp 15 years that underwent ECLS for cardiac surgery in our hospital. The only exclusion criterion was lack of signed informed consent.
One of the researchers (JMR), trained in transorbital sonography, performed all the ultrasound measurements of the ONSD.
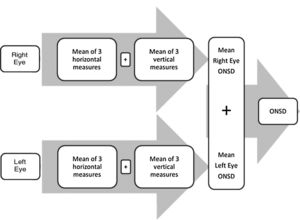
TechniqueWe analysed ONSD measurements at 3 time points: baseline (preoperative) and 6 and 24 h after surgery (postoperative). At the 6-h time point, all patients were being weaned from mechanical ventilation. At admission (baseline) and 24 h post surgery, all patients were awake and cooperative. At admission and after surgery, the ONSD was measured when haemodynamic parameters were most stable, with patients in the supine position and the head elevated to 30°. After applying coupling gel on the eyelid, the researcher obtained 3 horizontal and 3 vertical measurements for each eye with a 7−10 MHz linear transducer. The measurements were performed 3 mm behind the optic nerve papilla. We calculated the mean ONSD measurement for each eye, and then calculated the mean of the 2 eyes to use in the analysis (Fig. 1). We defined asymmetry as an interocular ONSD difference of 0.4 mm or greater.
Characteristics of the cardiac surgery proceduresAll procedures were elective and performed by the same surgical team: 1 paediatric cardiovascular anaesthesiologist, 1 paediatric interventional cardiologist and 2 paediatric cardiac surgeons. The anaesthesia protocol was adjusted based on the pulmonary blood flow (PBF) and weight of the patient. Per protocol, extubation was never performed in the operating room, but within 12 h from completion of the surgical procedure. All patients received neuromuscular blockade.
Extracorporeal life support was performed using an occlusive roller pump under hypothermic circulatory arrest (32–33 °C). All patients underwent haemodilution and received heparin in adherence with recommendations for paediatric perfusion. The interventional cardiologist maintained an appropriate blood flow rate per the established protocol (target mean arterial pressure of 40–70 mmHg).
We classified patients into 2 groups according to the indication for cardiac surgery: increased PBF and decreased PBF. We collected the following information from the operative reports: ECLS time, aortic cross-clamp time, intraoperative serum lactate levels.
Statistical analysisWe have expressed quantitative variables as median and interquartile range (IQR) based on the shape of the distribution determined by means of the Kolmogorov–Smirnov test, and categorical variables as percentages. In the univariate analysis, we compared groups using the Mann–Whiney U test for quantitative variables and the chi square test for categorical variables. We used the Wilcoxon signed-rank test to analyse differences between measurements made at different time points. Lastly, we calculated the Spearman correlation coefficient to assess the association of ONSD with intraoperative variables. We defined statistical significance as a p-value of less than 0.05. The analyses were performed with the software IBM SPSS Statistics version 20.0.
ResultsWe analysed data for 23 patients. Twelve were female (52.2%) and the median age was 14 months (IQR, 9–24 months). The median weight was 7.5 kg (IQR, 5.4–9.4 kg). Apart from 2 patients who died (mortality, 8.7%), all patients underwent the ONSD measurements established in the study protocol. Thus, the analysis included data for 21 patients for the 24 h post-surgery time point.
Congenital heart disease with increased PBF was the indication for surgery in 15 patients (65.2%). To classify the sample according to surgical risk, we used the Risk Adjustment for Congenital Heart Surgery 1 (RACHS-1) score. Based on the RACHS-1 scores, 10 patients (43.5%) were category I, 9 patients (39.1%) category II and 4 patients (17.4%) category III. There were no patients classified into the higher risk categories.
As for the time variables, the median ECLS time was 60 min (IQR, 33−102 min) and the median aortic cross-clamp time 32 min (IQR, 16−64 min). Lastly, the median serum lactate level was 1.4 mmol/L (IQR, 1.0–2.2 mmol/L). Table 1 compares these data in the PBF groups.
Baseline characteristics by type of congenital heart disease.
| Increased PBF | Decreased PBF | P | |
|---|---|---|---|
| n = 15 | n = 8 | ||
| Male sex, n (%) | 7 (63.6%) | 4 (36.4%) | .61a |
| Age, (months) | 17 (9–32) | 12.5 (9–18.5) | .21b |
| Weight, (kg) | 7.8 (5.5–11.0) | 7.5 (5.3–8.0) | .03b |
| RACHS-1 risk score, n (%) | |||
| I | 10 (100%) | – | .001c |
| II | – | 4 (44.4%) | |
| III | 5 (55.6%) | 4 (100%) | |
| ECLS time (min) | 49 (28−77) | 99 (75.7−138.5) | .007b |
| Aortic cross-clamp time (min) | 20 (16−43) | 51 (0.0−77.5) | .77b |
| Blood lactate (mmol/L) | 1.4 (0.90−1.70) | 2.0 (1.12−4.00) | .11b |
All values expressed as median (IQR) unless otherwise specified.
ECLS, extracorporeal life support; PBF, pulmonary blood flow; RACHS-1 = Risk Adjustment for Congenital Heart Surgery 1.
The median baseline ONSD was 3.1 mm (IQR, 2.9–3.6 mm). We found asymmetry in the ONSD measurements in only 3 patients: one with a larger diameter in the left eye (difference, 0.4 mm) and 2 with the larger diameter in the right eye (differences of 0.7 and 0.5 mm). We found a strong correlation between the ONSD measurement of the left and right eyes (r = 0.872, P < .001).
Relative to baseline, ONSD values had increased a median of 0.015 mm (IQR, –0.005 to 0.025 mm) at 6 h post surgery (P = .03), which resulted in a positive correlation between ECLS time and ONSD values at 6 h (Fig. 2), although the correlation was not statistically significant (P > .05). The ONSD values returned to baseline levels by 24 h post surgery, with a median change from baseline to 24 h of 0.31 mm (IQR, –0.055 to 0.78 mm). Table 2 shows the correlations coefficients obtained for the total sample and for each of the PBF groups. We found a statistically significant correlation between the ONSD and ECLS time for ONSD at 24 h (r = 0.476; P = .029) and the change in ONSD from 6 to 24 h (r = –0.538; P = .012) in the total sample, and for the change in ONSD from 6 to 24 h in the group of patients with increased PBF (r = –0.706; P = .003), but not in the group of decreased PBF (Table 2).
Correlation between cardiac surgery times, serum lactate and optic nerve sheath diameter.
| ONSD at 6 h | ONSD at 24 h | Δ ONSD Baseline—6 h | Δ ONSD Baseline—24 h | Δ ONSD 6 h–24 h | |
|---|---|---|---|---|---|
| Total sample | |||||
| ECLS time | 0.340 | 0.476* | 0.014 | –0.296 | –0.538* |
| Aortic cross–clamp time | –0.043 | 0.340 | 0.081 | 0.399 | –0.436* |
| Serum lactate | 0.151 | 0.096 | 0.025 | 0.074 | –0.310 |
| Increased PBF | |||||
| ECLS time | 0.108 | 0.453 | 0.029 | 0.370 | –0.706** |
| Aortic cross–clamp time | –0.072 | 0.326 | 0.189 | 0.214 | –0.593* |
| Serum lactate | –0.055 | 0.064 | –0.026 | 0.115 | –0.144 |
| Decreased PBF | |||||
| ECLS time | –0.261 | 0.377 | –0.048 | 0.377 | –0.464 |
| Aortic cross–clamp time | –0.253 | 0.235 | –0.037 | 0.471 | 0.088 |
| Serum lactate | 0.000 | 0.257 | –0.096 | 0.143 | –0.714 |
ECLS, extracorporeal life support; ONSD, optic nerve sheath diameter; PBF = pulmonary blood flow; Δ ONSD, difference between ONSD measurements.
All values correspond to Spearman’s rho.
Measurement of the ONSD is a relatively new application of the optic ultrasound examination that, unlike other imaging techniques such as CT or MRI, can be performed at the bedside.9 In paediatric patients that undergo cardiac surgery, the need for ECLS carries a risk of central nervous system injury,3 mainly due to brain oedema secondary to ischaemia and hypoxia.4 Previous studies have found a direct correlation between ECLS and aortic cross-clamp times and the duration of decreased cerebral perfusion.4,10 On the other hand, there is evidence in the medical literature of a strong correlation between ONSD and intracranial pressure (ICP).6,10 Thus, it follows that there must be an association between ECLS time and the ONSD. To our knowledge, this is the first study to demonstrate that this correlation does exist.
Having established this correlation, we are better situated to address further aspects of the relationship between these two variables, such as PBF. In our study, we found statistically significant differences in the baseline characteristics of the 2 the groups of participants established based on the PBF: age, weight, and surgical risk category. We found a positive correlation between the ONSD and both the ECLS and aortic cross-clamp times in patients with a high PBF, but only with the ECLS time in the total sample. Interestingly, we found a negative correlation between the changes in ONSD from 6 to 24 h and ECLS time, which can be interpreted as follows: the longer the duration of ECLS, the less difference there will be in ONSD values between measurements. We believe that this could mean that, independently of ONSD values at 6 h (which may be higher due to a longer duration of ECLS), there will be a tendency to maintain such ONSD values for as long as 24 h after surgery. If this finding is corroborated in future studies, for the purpose of clinical practice this would mean that ONSD remains stable through time, making abrupt changes in the postoperative period less likely.
Nevertheless, several issues must be addressed to correctly appraise the validity of our findings. First, even though the results of this study show a statistically significant correlation between ONSD values and ECLS time, the ONSD values were always within the normal range. In addition, none of the patients developed signs or symptoms of increased ICP. There is also the issue of age: most of the patients that require ECLS are infants (in this study, the median age was 17 months), which restricts the applicability of the results mainly to this age group. Second, previous studies have shown that the correlation between ONSD and ICP is not linear11 and is susceptible to intraindividual variation, and therefore ECLS time is just one of several factors that influence ONSD in the patient. Future studies need to include additional factors, such as the patency of the anterior fontanelle10 and the ultrastructure of subarachnoid space.12 Third, due to the dynamic nature of the ONSD-ICP correlation, the timing of the measurements is another factor on which further research is required to determine the optimal protocols. In our study, we took measurements at 6 and 24 h post surgery, but we recognize that other time frames are also valid, and that more frequent measurements may need to be made in case of longer ECLS times. Eventually, we will be able to determine the standard responses of ONSD to various times. These issues significatively reduce the clinical relevance of the results. Finally, although we calculated the sample size based on the expected correlation and managed to recruit the full necessary sample size, it could be argued a separate sample size calculation should be made for each of the PBF groups. Therefore, we plan to address this issue in future studies, making separate sample size calculations for each subset of patients.
ConclusionOur study in a sample of paediatric patients undergoing cardiac surgery with ECLS found a positive correlation between ONSD measured at 24 h post surgery and ECLS time. The ONSD had increased within the normal range at 6 h post surgery and returned to baseline after 24 h. Variations in the ONSD are observed even in patients without signs or symptoms of increased ICP. Further research is required to identify the factors related to the effect of ECLS time on the ONSD and to the variations in the ONSD.
Please cite this article as: Rivas-Rangel J, García-Arellano M, Marquez-Romero JM. Correlación del diámetro de la vaina del nervio óptico y el tiempo de circulación extracorpórea. An Pediatr (Barc). 2022;96:91–96.