The ventilation rates and tidal volumes (VT) delivered by the conventional ventilatory modalities lack the spontaneous variability characteristic of physiological rhythms, which are believed to be protective for the lung.1 Experimental studies have shown that compared to “monotonous” modalities, variable ventilation results in an improved distribution of VT, greater lung recruitment and increased production of surfactant,2 improving the ventilation/perfusion ratio, gas exchange and lung mechanics while reducing the production of proinflammatory cytokines, pulmonary oedema and ventilator-associated lung injury.3 Our aim was to establish whether neurally adjusted ventilatory assist (NAVA) increases breath-to-breath variability and decreases patient-ventilator asynchrony compared to pressure-regulated volume control (PRVC) ventilation.
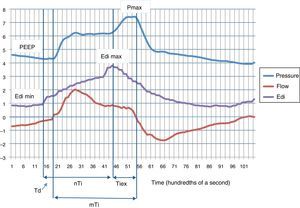
We present the case of one neonate born preterm at 36 weeks’ gestation with a birth weight of 2000g that received mechanical ventilation due to extrapulmonary problems with a Servo-n® (Maquet, Solna, Sweden) and an Edi catheter for detecting diaphragmatic electrical activity (initial support with PRVC: VT 5mL/kg; pneumatic trigger, 0.3mL; positive end-expiratory pressure [PEEP], 4cmH2O; FiO2, 0.21; backup respiratory rate [RR], 20bpm). While the patient was awake, calm and stable, we recorded neural respiratory values (peak Edi, minimum Edi, neural inspiratory time [nTi]) and mechanical respiratory values (Pmax, mechanical inspiratory time [mTi], VT) over a period of 30s (Fig. 1). Later on, after obtaining verbal consent from the mother, the patient was switched to NAVA mode: NAVA level 1; Edi trigger, 0.5μV; PEEP, 4cmH2O; FiO2, 0.21; and apnea time for backup ventilation, 5s. After a few minutes of adaptation, the same parameters were recorded for 30 seconds again. We also calculated the trigger delay (Td, time elapsed between initial increase in Edi and the beginning of positive inspiratory flow) and the inspiratory time in excess (Tiex, time elapsed between the peak Edi and the beginning of expiratory flow) for each respiratory cycle. The asynchrony index (AI) was calculated as the total number of major asynchrony events (autotriggering, double-triggering and wasted neural efforts) divided by sum of the ventilator RR plus the number of ineffective efforts, multiplied by 100. The breath-to-breath variability was calculated as the coefficient of variation (CV=standard deviation [SD]/mean) for each of the parameters. Results are expressed as mean and SD, and compared them by means of the Mann–Whitney U test, defining statistical significance as P<.05.
We analysed 21 respiratory cycles under PRVC and 35 under NAVA. Table 1 shows the comparison of neural and mechanical respiratory variables. We observed three major asynchrony events under PRVC (AI, 14.3%) and none under NAVA. After switching to NAVA, the CVs increased: peak Edi, from 0.46 to 0.55; Pmax, from 0.25 to 0.35; mTi, from 0.13 to 0.32; VT, from 0.27 to 0.65; and we observed a reduction in Td and Tiex (Table 1).
Respiratory variables.
| Variablesa | PRVC (n=21) | NAVA (n=35) | P |
|---|---|---|---|
| nTi (ms) | 405±133 | 358±118 | .190 |
| mTi (ms) | 440±55 | 340±108 | <.001 |
| Td (ms) | 77.8±68.8 | 45.8±32.9 | .106 |
| Td>100ms (%) | 22 | 6 | .113 |
| Edimax (μV) | 3.5±1.6 | 6.6±3.6 | .001 |
| Edimin (μV) | 0.4±0.2 | 0.9±0.4 | <.001 |
| Pmax (cmH2O) | 10.3±2.6 | 10.9±3.8 | .602 |
| VT (mL) | 10.2±2.8 | 9.1±5.9 | .435 |
| Tiex (ms) | 118±93 | 74±22 | .082 |
This is the first study conducted in Spain that compares respiratory variability in two modes of assisted mechanical ventilation in a newborn. Our main finding was that NAVA increased the variability of nearly every parameter under study. We also observed a significant decrease in the AI compared to PRVC.
Most modern neonatal ventilators offer flow synchronisation and some kind of volume guarantee that is established by the professional based on clinical and gasometric criteria. Few studies have assessed the efficacy of flow synchronisation in the neonate. There is evidence, however, that NAVA significantly improves synchrony in newborns, children and adults.4 Synchronisation by Edi reduces Td, and allows the patient to adjust the Ti, RR and VT in real time, as the brainstem respiratory centres continuously receive information from central and peripheral chemoreceptors, thoracic and diaphragmatic mechanoreceptors, and stretch and irritant receptors in the lungs and respiratory airways.5
Decreased respiratory variability during weaning has been associated with extubation failure, and asynchronies with prolonged mechanical ventilation, discomfort, sleep fragmentation, etc.6 Although few studies have been conducted in humans, currently available data suggest that increased variability in mechanical ventilation could have beneficial effects for patients. NAVA increases this variability and improves patient-ventilator synchrony, bringing mechanical ventilation closer to natural ventilation. Prospective, randomised and well-designed studies including patients with different types of pulmonary disease are required to demonstrate the potential of this modality of respiratory support to improve clinical outcomes and patient prognosis in the long run.
Please cite this article as: García-Muñoz Rodrigo F, Urquía Marti L, Galán Henríquez G, Rivero Rodríguez S, Siles Quesada C. La ventilación asistida ajustada neuralmente incrementa la variabilidad respiración a respiración y mejora la sincronización paciente-ventilador. An Pediatr (Barc). 2016;84:231–232.