Asthma is one of the main chronic diseases in childhood, due to its high prevalence and its social and health costs. This document is a summary of a consensus guideline approved by 6 Spanish pediatric societies related to asthma and endorsed by the Spanish Pediatric Association. Asthma is easily identifiable by clinical criteria in most patients. Spirometry and other tests are helpful for diagnosis, especially in atypical cases. Asthma exacerbation is a frequent manifestation of the disease and must be identified and treated promptly. When asthma symptoms are frequent and the quality of life is affected, maintenance treatment must be instituted to achieve control of the disease. Low-dose inhaled corticosteroids are effective and safe for long-term use. Education of the patient with asthma is essential for good control. The main reason for poor asthma control is non-compliance with treatment, either due to its erratic and insufficient administration, or due to poor application technique of inhaled drugs. If control is not obtained despite adequate treatment, the diagnosis must be reconsidered, as well as the factors or comorbidities that make control difficult. Other drugs can be added to avoid high doses of inhaled corticosteroids, notably montelukast or long-acting β2 adrenergic agonists. Severe or difficult-to-control asthma, which does not respond to the usual treatments, should be managed in specialized units.
El asma es una de las principales enfermedades crónicas de la infancia, por su elevada prevalencia y por su coste sociosanitario. Este artículo es un resumen de la guía de consenso alcanzada por 6 sociedades pediátricas relacionadas con el asma y avalada por la Asociación Española de Pediatría. El asma es fácilmente identificable por criterios clínicos en la mayoría de los pacientes. La espirometría y otras pruebas son de gran ayuda, especialmente en los casos atípicos. La crisis de asma es una manifestación frecuente de la enfermedad y debe ser identificada y tratada con prontitud. Cuando los síntomas de asma son frecuentes y afectan a la calidad de vida es preciso instaurar un tratamiento de mantenimiento para conseguir el control de la enfermedad. Los glucocorticoides inhalados a dosis bajas son eficaces y seguros para su uso prolongado. La educación del paciente con asma es esencial para obtener un buen control. El principal motivo de mal control del asma es el incumplimiento del tratamiento, ya sea por su administración errática e insuficiente, o por la mala técnica de administración de los fármacos inhalados. Si no se obtiene el control pese a un tratamiento adecuado es preciso reconsiderar el diagnóstico, así como los factores o comorbilidades que dificultan el control. Se pueden añadir otros fármacos para evitar las dosis altas de los glucocorticoides inhalados, principalmente el montelukast o los agonistas β2 adrenérgicos de acción prolongada. El asma grave o de difícil control, que no responde a las medidas habituales, debe ser atendida en unidades especializadas.
Asthma is one of the most prevalent chronic diseases of childhood and results in a significant decrease in the quality of life of patients and their families as well as significant economic costs for both individuals and society.1 Asthma usually has onset in childhood and the prevalence in the pediatric population of Spain is estimated at 10%, although it varies between regions and age groups.2 This article summarises the consensus document developed by 6 pediatric societies with an interest in asthma (SEICAP, SENP, SEPEAP, AEPAP, SEUP and AEEP), endorsed by the Asociación Española de Pediatría (Spanish Association of Pediatrics), which we recommend consulting, as it includes a substantial amount of practical information and explains in detail some important aspects that did not fit in this article.3 The consensus document was developed with the aim of bringing the essential knowledge on asthma in children and adolescents in Spain up to date, prioritising the development of tables and figures in the most simplified and summarised form possible.
Definition and diagnosis of asthmaDefinition of asthmaAsthma is a disease with a heterogeneous presentation characterised by a series of clinical findings that reflect the presence of chronic, diffuse and fluctuating or reversible obstruction of the lower respiratory tract, generally of an inflammatory nature. Thus, it is a clinical diagnosis that refers to patients with certain manifestations that do not have a well-established cause.4 Different aetiopathogenic factors, both genetic and environmental, have been associated with asthma, a combination of which is found in each patient, causing bronchial inflammation and hyperresponsiveness. Asthma can be diagnosed from the early years of life.5 Although it is a chronic disease, it may improve spontaneously in many affected children whose symptoms seem to resolve as they grow, but predicting the course of disease at the individual level is challenging. A greater severity of asthma and sensitization to allergens in the early years are some of the risk factors for persistent asthma at a later age.
Clinical manifestationsThe symptoms of asthma may be very mild or even absent while the disease is stable. Cough is a common and early symptom, but also nonspecific. If the obstruction increases, tachypnoea and shortness of breath develop, with a prolonged expiration and use of accessory muscles. Breathing starts to make sound, and wheezing is the most characteristic sound associated with asthma. Older children may experience dyspnoea and chest pain or tightness. Asthma exacerbations are often preceded and triggered by upper respiratory tract infections. Exacerbations with a sudden onset (episode of bronchospasm) are more common in older children and in allergic asthma.
Diagnosis of asthmaThe initial diagnosis in children is essentially clinical and based on the presence of all 3 criteria listed in Table 1. Nonresponse to treatment or the presence of atypical features should prompt reassessment of the diagnosis (Table 2). Lung function tests play an important role in establishing the presence of airflow obstruction and its lability, especially in atypical or uncertain cases. Nevertheless, there are potential limitations that may preclude the use of spirometry and must be taken into account:
- •
Technical requirements concerning the equipment, reference values and the professional performing the test.
- •
It may be difficult to gain the cooperation of some children and, in very young children, impossible.
- •
Values may be normal in many patients with asthma when they are not experiencing an exacerbation.
- •
Lung function abnormalities may be indicative of respiratory problems other than asthma.
Criteria for diagnosis of asthma in pediatric patients.
| All 3 criteria must be metSymptoms of bronchial obstruction (acute and recurrent, or persistent and fluctuating), witnessed by a clinician, with the clinical manifestations detailed in the text |
| The symptoms exhibit pronounced variation, spontaneously or with asthma treatment |
| The above symptoms cannot be fully explained by other diseases that have a similar presentation (Table 2) |
| In addition, whenever possible, variable airflow obstruction will be objectively confirmed |
Differential diagnosis of asthma.
| Recurrent upper respiratory tract infection: recurrent rhinitis, sinusitis, adenoiditis and laryngitis |
| Bronchopulmonary dysplasia or chronic lung disease of prematurrity |
| Anatomical airway malformations: pulmonary, oesophageal or vascular |
| Tracheobronchial malacia or stenosis |
| Mediastinal masses, adenopathy (tuberculosis), tumours |
| Foreign body airway obstruction |
| Dysphagia, gastro-oesophageal reflux and aspiration syndrome |
| Non-recurrent bronchiolitis or bronchitis |
| Chronic lung disease: cystic fibrosis, bronchiolitis obliterans, primary ciliary dyskinesia, interstitial lung disease, bronchiectasis, etc. |
| Inducible laryngeal obstruction (mainly vocal cord dysfunction) |
| Psychogenic cough and respiratory disorders |
| Heart disease |
| Immunodeficiencies |
Thus, spirometry must be interpreted accurately in the context of the information available on the patient, and cannot be used in isolation to confirm or rule out the presence of asthma.6 Other techniques may be used to support the diagnosis of asthma, such as lability of the peak expiratory flow rate or the fractional exhaled nitric oxide. An allergy evaluation is useful in the overall assessment of the patient, but it is not required to make the diagnosis, which is mainly based on the findings of the clinical evaluation supplemented, whenever possible, by lung function tests.
Asthma exacerbationsIdentification of asthma exacerbationsAn exacerbation or attack is an episode in which symptoms of asthma develop or worsen. Early identification and treatment of exacerbations are key priorities. A history-taking and physical examination are usually sufficient to make the diagnosis. Exacerbations usually develop in patients with a previous diagnosis of asthma, but they may also be the presentation at onset. The differential diagnosis is usually made in the first few episodes, most frequently in early childhood (Table 2). Patients and their families must be able to recognise and adequately manage asthma exacerbations.
Initial evaluation of asthma exacerbationsIt is essential that the severity of the episode is assessed based on the findings of the physical examination combined with the risk factors identified in the history-taking (Table 3). The severity of an exacerbation can be established quickly and pragmatically based on clinical criteria, which may be supplemented by pulse oximetry (Table 4). Breathing sounds on auscultation, which may be florid or quiet, are not strongly correlated to severity. The response to treatment is an important factor. Diagnostic tests are reserved for cases of severe asthma, with a protracted course or with an uncertain diagnosis.
Comorbidities and other factors that may hinder asthma control and increase the risk of severe asthma exacerbations.
| History indicative of severe or poorly controlled asthma |
| History of severe exacerbations, rapid worsening, admission to PICU |
| Several hospital admissions or visits to emergency department in the past year |
| Visit(s) to emergency department in the past month |
| Frequent use of bronchodilators |
| Limitation of activity |
| Inadequate or insufficient treatment |
| Inadequate follow-up |
| Nonadherence to treatment |
| Poor inhaler technique |
| Family and social problems |
| Family conflict |
| Poverty |
| Marginalization |
| Environmental or home pollution |
| Psychological or psychiatric problems |
| Difficulty accessing health care resources and emergency care |
| Age-related factors |
| Early years of life: small airway calibre, frequent respiratory infections, fewer available and less effective drugs for asthma |
| Adolescence: rebelliousness, loss to follow-up and decline in adherence, decreased risk perception, abuse of short-acting bronchodilators |
| Spirometry features indicative of obstruction or lability |
| Decreased FEV1 or FEV1/FVC |
| Positive bronchodilator responsiveness test |
| Excessive lability of peak expiratory flow |
| Other allergies and features of atopy |
| Polysensitization |
| Allergic rhinitis or chronic rhinosinusitis |
| Food allergy |
| Frequent or continuous exposure to allergens relevant to the patient |
| Main comorbidities that hinder asthma control |
| History of preterm birth and bronchopulmonary dysplasia |
| Passive or active smoking |
| Chronic neurologic or neuromuscular disease |
| Obesity |
| Sleep apnoea-hypopnoea syndrome |
| Functional or psychogenic disorders with an impact on respiration, including vocal cord dysfunction and psychogenic hyperventilation, cough or dyspnoea |
FEV1, forced expiratory volume in the first second; FVC, forced vital capacity; PICU, pediatric intensive care unit.
Assessment of asthma severity through the direct examination of the patient or through the pulmonary score and pulse oximetry.
| 1)Summary of the classification of the severity of the exacerbation based on the examination of the patient | ||||||
| Mild exacerbation: asthma signs and symptoms with absent or mild increase in work of breathing | ||||||
| Moderate exacerbation: marked increase in work of breathing without significant change in the general appearance and behaviour of the patient | ||||||
| Severe exacerbation: marked increase in work of breathing that alters the appearance and behaviour of the patient (subjective sensation of tightness of throat): irritability or somnolence, changes in voice, air hunger, increased comfort in seated positions, tachypnoea, and severe chest retractions or slow and laboured breathing with severe hypoventilation | ||||||
| 2) Integrated assessment with the pulmonary score and pulse oximetry to determine the severity of the exacerbation | ||||||
| A) Pulmonary score | ||||||
| Score | Respiratory rate (bpm) | Wheezinga | Use of accessory muscles | |||
| <6 years | ≥6 years | |||||
| 0 | <30 | <20 | No | No | ||
| 1 | 31–45 | 21–35 | Terminal expiration (with stethoscope) | Mild increase | ||
| 2 | 46–60 | 36–50 | Entire expiration (with stethoscope) | Increase | ||
| 3 | >60 | >50 | Both inspiration and expiration, (without stethoscope) | Maximum activity | ||
| B) Global assessment of severity integrating the pulmonary score and oxygen saturation measured by pulse oximetry | ||||||
| Pulmonary Score | SpO2 % | |||||
| Mild | 0–3 | >94 | ||||
| Moderate | 4–6 | 91–94 | ||||
| Severe | 7–9 | <91 | ||||
SpO2, oxygen saturation measured by pulse oximetry.
Each section has a score ranging from 0 to 3 (total minimum 0, maximum 9). Mild exacerbation: 0–3 points; moderate, 4–6 points; severe, 7–9 points.
In case of disagreement between the clinical score and the oxygen saturation, the greatest severity is assigned.
Treatment is contingent on severity (Fig. 1). Table 5 presents the dosage of asthma medications. Salbutamol should preferably be administered with a pressurized metered-dose inhaler with a spacer (in children aged less than 4 years, through a face mask), even during severe attacks.7,8 Nebulization may be contemplated in special situations. Systemic corticosteroids, usually delivered orally,9 are indicated in moderate to severe exacerbations or when there is risk of worsening (Table 3). There is no conclusive evidence that increasing the dose of inhaled corticosteroids (ICSs) during an exacerbation can improve outcomes in children.10 Ipratropium bromide is used in combination with salbutamol to manage moderate to severe exacerbations. Intravenous magnesium sulphate seems effective and safe for treatment of severe asthma exacerbations.11 Oxygen should be administered as needed to maintain an oxygen saturation greater than 92%–94%. In case of mild exacerbation or favourable response, treatment can continue at home. Otherwise, the patient needs to be treated in the hospital.
Simplified algorithm of the initial management of asthma exacerbations based on severity and response to treatment. For details about medication dosage, see Table 5.
ICU, intensive care unit; SABA, short-acting β2 receptor agonists (salbutamol and terbutaline); SaO2, oxygen saturation.
*Corticosteroids may be used earlier in children with allergic asthma and risk factors, but their indication for management of non-allergic asthma in early childhood is still under debate.
**Supplemental oxygen may also be needed in nonsevere exacerbations to maintain an adequate oxygen saturation.
Recommended dosage of the main drugs used to manage asthma exacerbations.
| Drug | Administration | Dose |
|---|---|---|
| Salbutamol | Suspension for inhalation in pressurised canister: 100 μg per puff (with spacer) | Mild exacerbation: 2−4 puffsModerate-severe exacerbation: 4−10 puffsMay be repeated every 20 min based on response |
| Powder for inhalation:-Novolizer®: 100 μg/dose-Clickhaler®: 90 μg/dose | Mild exacerbation: 1−2 actuationsModerate-severe exacerbation: not recommended | |
| Solution for nebulization:-Ventolin® solution for nebulization: 5 mg/mL-Salbuair® ampoules of solution for nebulization:2.5 mg/2.5 mL (1 mg/mL)5 mg/2.5 mL (2 mg/mL) | - Fixed dose: 2.5−5 mg- Based on weight: 0.15 mg/kg (minimum 2 mg-maximum 5 mg)In both cases, prepare to the volume required by the nebulizer, usually 4−5 mL, adding isotonic saline if neededMay be repeated every 20 min based on response | |
| Terbutaline | Powder for inhalation (Turbuhaler®): 500 μg/dose | Mild exacerbation: 1−2 actuationsModerate-severe exacerbation: not recommended |
| Prednisone, prednisolone | OralParenteral in severe cases | 0.5−2 mg/kg/day (maximum 60 mg/day) in 1−2 doses per day. Can be maintained for up to 3−5 days |
| Dexamethasone | OralParenteral in severe cases | 0.3−0.6 mg/kg/day(maximum 12 mg) for 1−2 days |
| Ipratropium bromide | Suspension for inhalation in pressurised canister: 20 μg per actuation (with holding chamber) | Only in moderate-severe exacerbations: 2−8 actuations (40−160 μg). May be repeated every 20 min combined with salbutamol until the exacerbation is no longer severe |
| Solution for nebulization in monodose vials of 250 and 500 μg | Only in moderate-severe exacerbations:- Age < 6 years: 125−250 μg- Age 6−12 years: 250−500 μg- Age > 12 years: 500 μgAdminister in combination with salbutamol until the exacerbation is no longer severe |
The goal of treatment is to maintain the child asymptomatic or with the minimum possible symptoms and exacerbations, allowing a life without limitations in daily activity. With adequate treatment, asthma can be controlled completely or almost completely in most cases. Treatment is based on the cooperation of patients and health care professional and must be individualised and adjusted on an ongoing basis based on the aetiology and severity of asthma and the response to treatment.
Inhaled corticosteroids are the first-line treatment for asthma control on account of their efficacy and adequate therapeutic index, especially for allergic asthma. Low to medium doses (200−400 μg of budesonide or 100−200 μg of fluticasone propionate a day) usually suffice. If treatment does not achieve adequate control and adherence and correct inhaler technique are verified, it is more effective to add a second drug than to increase the ICS dose further. Montelukast can be used as monotherapy in nonsevere asthma or combined with ICSs and other drugs in uncontrolled asthma, and administration in a single dose by the oral route facilitates adherence. However, its efficacy is lesser and less predictable compared to ICSs.12 Furthermore, adverse effects have been reported in association with its use, including sleep disturbances, abdominal pain and neuropsychiatric disorders.13 Long-acting β2 agonists (LABA) are authorised for use in children aged 4 years and older. They must always be given in combination with an ICS in case of poor control to avoid high doses of ICSs. Tiotropium bromide is added to the regimen of patients aged 6 years and older with poor asthma control despite treatment with ICSs combined with other drugs. The use of other drugs, such as chromones, theophylline or macrolides, is decreasing or only prescribed in exceptional cases.
Stepwise asthma treatment and controlCurrent guidelines recommend a stepwise approach to treatment in which the selection of drugs and increases or decreases in the dose of ICSs are based on disease severity and the response to treatment, while monitoring correct adherence on an ongoing basis. Asthma is classified based on the impairment experienced by the patient and the treatment required to achieve adequate control (Table 6). When asthma symptoms have been under control for 2–3 months, it is possible to step-downasthma medications with the aim of identifying the minimum effective treatment. If an adequate response is not achieved or the previous control is lost, an evaluation should be performed before going up a step:
- •
Confirm the diagnosis of asthma or consider alternative diagnoses (Table 2).
- •
Assess adherence to treatment.
- •
Assess inhaler technique.
- •
Assess the measures taken to avoid exposure to noxious agents (allergens, tobacco smoke or other pollutants).
- •
Assess for the presence or development of risk factors and comorbidities (Table 3).
- •
Provide or revise the asthma written plan.
Summary of the classification of asthma and the measures required for its management.
| Asthma severity and control | ||||
|---|---|---|---|---|
| Episodic (mild) | Persistent (moderate) | Poorly controlled (severe) | ||
| Clinical characteristics | Exacerbations | Infrequent and mild | Frequent or not mild | Frequent and severe |
| Symptoms between exacerbations | Absent | Occasional | Frequent | |
| Exercise capacity | Good | Fair | Poor | |
| Asthma-related quality of life | Good | Fair | Poor | |
| Bronchodilator use | Occasional | Frequent | Very frequent | |
| Lung function | Normal | Slightly altered | Very altered | |
| Treatment and follow-up | Maintenance or first-line treatment (minimum) | Not needed | Low-dose ICS or montelukast | Medium-dose ICS + LABA or SMART |
| Alternative treatments (additional) | Low-dose ICS or montelukast in periods of poorer control | Low-dose ICS + LABAMedium-dose ICS ICS + montelukastSMART in adolescents | Combination of several drugs, including tiotropiumHigh-dose ICSBiologic agentsOther drugs or measures used infrequently or the efficacy of which has not been well established | |
| Relief therapy | As-needed SABA delivered with method appropriate for age. Consider formoterol + budesonide in adolescents | |||
| Common aspects (intensification as severity increases) | Asthma education. Avoidance of triggers. Investigation of alternative diagnoses, risk factors and comorbidities. Assessment of adherence and administration technique. Asthma action plan, preferably in writing | |||
| Allergy testing | In case of presentation indicative of allergy or association with allergic rhinitis | Generally recommended | Allergy testing and lung evaluation by specialist | |
ICS, inhaled corticosteroids; LABA, long-acting β2 agonists; SABA, short-acting β2 agonists; SMART, single maintenance and reliever therapy with formoterol + budesonide combined in a single inhaler.
In early childhood, asthma tends to manifest with frequent exacerbations associated with upper respiratory tract infections. In these early years, the only drugs available for maintenance therapy are ICSs and montelukast, and their efficacy is lesser compared to later in life. A possible option in preschool-age children that are not receiving ICS therapy for maintenance is administration of an ICS at a high dose for a short period of time (7–10 days) at the onset of symptoms of respiratory infection to try to reduce the risk of a moderate to severe exacerbation. Starting from age 4 years, a LABA can be added to the ICS, and from age 6 years it is possible to use most of the available asthma medications in addition to dry powder inhalers. In adolescents, starting from age 12 years, the treatment options are the same available for adults, while the autonomy of the patient also increases. In adolescence, symptoms tend to be milder and more continuous, and abuse of short-acting β2 agonists (SABAs) and poor adherence to maintenance treatment are common, which may result in poorer asthma control and an increased risk of severe exacerbations. One treatment option in this age group is what is known as single maintenance and reliever therapy (SMART). This strategy consists in the administration of formoterol (a fast-onset LABA) combined with an ICS (budesonide) in a single inhaler. These devices are used for both maintenance therapy (1−2 doses a day) and relief therapy (as needed, up to a daily maximum of 56 μg of formoterol), which allows the patient to adjust the dose of ICS required for relief autonomously. This may be the most suitable option for treatment of poorly controlled asthma.14 The SMART approach cannot be used if the ICS is combined with a LABA other than formoterol, in which case the quick-relief drug should be a SABA. Table 7 summarises specific aspects of treatment at different ages.
Summary of specific clinical characteristics of asthma and approaches to treatment at different ages.
| Age < 4 years | Age 4−11 years | Age > 12 years | |
|---|---|---|---|
| Predominant type of exacerbation | Exacerbations triggered by viral infections | Exacerbations triggered by viral infections, allergens and other factors (e.g. physical activity) | Exacerbations triggered by viral infections, allergens and other factors (e.g. physical activity) |
| Symptoms between exacerbations | Infrequent | Slightly more frequent | Progressive increase in frequency. Decreased frequency of exacerbations |
| Preferred inhalation device | Holding chamber with oronasal mask | Holding chamber without a mask. Dry powder inhaler in older children | Dry powder inhaler |
| Inhaled corticosteroids | Fluticasone propionateBudesonide | Fluticasone propionateBudesonideBeclomethasonea | All, including mometasone, ciclesonide and fluticasone furoate |
| Other drugs for maintenance therapy | Montelukast | MontelukastLABATiotropium | All available to adultsSMART |
| Biologic agents | None | OmalizumabMepolizumab | OmalizumabMepolizumabDupilumabb |
LABA, long-acting β2 agonists; SMART, single maintenance and reliever therapy with formoterol and inhaled corticosteroids combined in a single inhaler.
Immunotherapy may improve quality of life and reduce the need for asthma medication in select patients considered eligible based on evidence demonstrating the clinical relevance of a given allergen in the evaluation.15 It may be particularly indicated in patients with concomitant allergic rhinitis.
Asthma educationPatient education is one of the main pillars of the management of asthma. All professionals that care for patients with asthma should be involved in the delivery of asthma education.16–18 This is an interactive and individualised process aimed at improving asthma control and the quality of life of patients and families. The importance of the role played by the child increases with age and peaks in adolescence, pursuing the maximum possible involvement of the patient in self-management. Providing written material on asthma education contents, especially the personalized asthma action plan, is helpful to patients and families.
The contents of educational interventions include basic knowledge of the disease, its triggers and its management. The goals include acquisition of knowledge and skills for delivery of the prescribed inhaled medication, avoidance of nonspecific asthma triggers (active and passive smoking, environmental pollution, cold and dry air, viral infections) as well as specific triggers (allergens). Regular physical activity and adequate nutrition are also important to avoid excess weight and sedentary lifestyles, which contribute to poorer asthma control. Certain health care staff should be trained to deliver asthma education (nurses, doctors, pharmacists).
The age of the patient is crucial for the purpose selecting the device used for inhalation, but what is most important is ensuring that the child is able to use the inhaler correctly (Table 8). The main reason asthma control is lost is lack of adherence. Inhaler technique must be reassessed at regular intervals.
Selection of the inhalation device based on age.
| Age | Device |
|---|---|
| <4 years | Pressurised inhalers with a valved holding chamber and delivery through an oronasal maskThe mask must create a tight seal so the negative inspiratory force is only exerted on the inside of the chamberThe child will take 5–10 easy breaths while inhaling the medication |
| 4–6 years | Pressurised inhalers with a valved holding chamber without use of a maskThe child will take 5–10 easy breaths while inhaling the medication; attempt deep inspirations to better inhale the drug |
| >6 years | Dry powder inhalers may be used if the patient demonstrates adequate techniqueMore portable, used without a spacerDifficult to use in case of severe-moderate obstruction (exacerbation) |
The choice of device may depend on the specific circumstances and preferences of the patient and the family. In every case, the clinician must ensure that the patient understands the technique and can execute it correctly.
Nebulizers should only be used in special cases or situations.
Most patients can achieve adequate control with conventional treatment. The main reason for poor asthma control is nonadherence to treatment, due to either noncompliance or poor inhaler technique. In other cases, asthma is difficult to control despite adequate adherence, and this is what constitutes severe asthma, which is found in as many as 5%–10% of asthmatic children. Asthma is considered severe when it results in substantial impairment in quality of life despite adequate adherence to treatment, or when its control requires treatments that may have significant adverse effects. The use of systemic corticosteroids for maintenance therapy is rare in paediatrics, as it is best to use these drugs for short periods of time as a temporary measure. Biologic agents are a last-resort option in patients that respond poorly to treatment. In school-age children and adolescents, the allergic phenotype is the most frequent form of severe asthma. Severe asthma is more difficult to define in infancy and early childhood, when there is a predominance of exacerbations triggered by respiratory infections that tend to become less frequent with age.
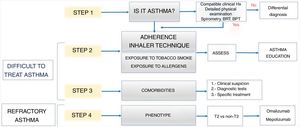
Initial management of patients with severe asthmaWe recommend a stepwise evaluation of patients with severe asthma19–21 (Fig. 2). Many of these patients need to be managed in specialised units if adequate control is not achieved.
- •
Step 1: Is it asthma? Some of the patients that receive care for potential severe asthma may have an alternate condition (Table 2). Special or invasive tests may be required.
- •
Step 2: Are there factors that hinder asthma control? Assess for potentially modifiable factors (Table 3). The most frequent are poor adherence to asthma medication or poor inhaler technique.22 Poor adherence is a particularly important problem in adolescence. It is also important to assess for the potential exposure to noxious substances, such as allergens, tobacco smoke and other pollutants.
- •
Step 3: Are there comorbidities that hinder asthma control? There may be concurrent diseases that, unless diagnosed and managed correctly, may hinder or preclude asthma control (Table 3).
- •
Step 4: What form of asthma does the patient have? This is usually investigated in specialty clinics. The most frequent phenotype in the paediatric age group is allergic. The non-allergic eosinophilic and neutrophilic phenotypes are less frequent.
Most patients can improve following a stepwise evaluation. Otherwise, the patient must be managed in a specialised unit and biologic therapy should be contemplated. The 2 biologic agents authorised for paediatric use in Spain are omalizumab (a monoclonal antibody that blocks IgE) and mepolizumab (a monoclonal antibody that targets eosinophils).
Asthma and COVID-19Children and adolescents infected by SARS-CoV-2 tend to be asymptomatic or developed mild symptoms of coronavirus disease 2019 (COVID-19). Asthma does not appear to be a risk factor for COVID-19, even in adult patients. The use of ICSs is not associated with an increased risk of developing the disease and it has been hypothesised that it may even have a protective effect. The consensus document details the main recommendations for the management and treatment of asthma during the COVID-19 pandemic.3
FundingThis project has not received any external funding.
Conflicts of interestLuis Moral has received fees or financial support for participation in educational activities, as a consultant or to attend scientific activities from Hero España, Merck-Allergopharma, ALK-Abelló, Reig Jofre, Leti, Faes Farma, Novartis, Inmunotek, GSK, and Alter.
Maite Asensi has received fees or financial support for participation in educational activities, as a consultant or to attend scientific activities from Nestlé, Ordesa, AstraZeneca, GSK, Pfizer and MSD.
Juan Carlos Juliá has received fees or financial support for participation in educational activities, as a consultant or to attend scientific activities from Hero España, Nestlé, Ordesa, Leti, GSK, Pfizer and MSD.
Cristina Ortega Casanueva has received fees or financial support for participation in educational activities, as a consultant or to attend scientific activities from ALK-Abelló, Reig Jofre, Leti and Diater.
José Sanz Ortega has received fees or financial support for participation in educational activities, as a consultant or to attend scientific activities from GSK, MSD and ALK-Abelló.
Laura Valdesoiro Navarrete has received fees or financial support for participation in educational activities, as a consultant or to attend scientific activities from GSK, Novartis, Leti and Reig Jofre.
José Valverde-Molina has received fees or financial support for participation in educational activities, as a consultant or to attend scientific activities from Novartis, GSK, AstraZeneca and TEVA.
The rest of the authors have no conflicts of interest to declare.
Please cite this article as: Moral L, Asensi Monzó M, Juliá Benito JC, Ortega Casanueva C, Paniagua Calzón NM, Pérez García MI, et al., Asma en pediatría: consenso REGAP, Anales de Pediatría. 2021;95:125.
Red Española de Grupos de Asma en Pediatría (Spanish Network of Working Groups on Paediatric Asthma).