The treatment of lung metastases of solid paediatric tumours and its impact on survival has been a controversial topic. The main studies on the subject have gathered data from patients with a variety of tumours such as Wilms’ tumour, Ewing sarcoma, rhabdomyosarcoma or synovial sarcoma. Many have focused on a specific type of tumour, without predicting significant differences among them. Moreover, whole lung irradiation (WLI) has been integrated in current international protocols despite the lack of evidence on its impact on overall survival (OS).
We conducted a retrospective study in a sample of 24 children with lung metastases treated between 2000 and 2017, of who 18 received WLI. We analysed clinical characteristics (Table 1), the outcomes of lung metastases, the response to chemotherapy or irradiation, the time to relapse, radiation doses and dose fractionation, and survival outcomes. Our objective was to establish the benefits of WLI in patients that received prophylactic irradiation and patients that received radical radiotherapy (Table 2).
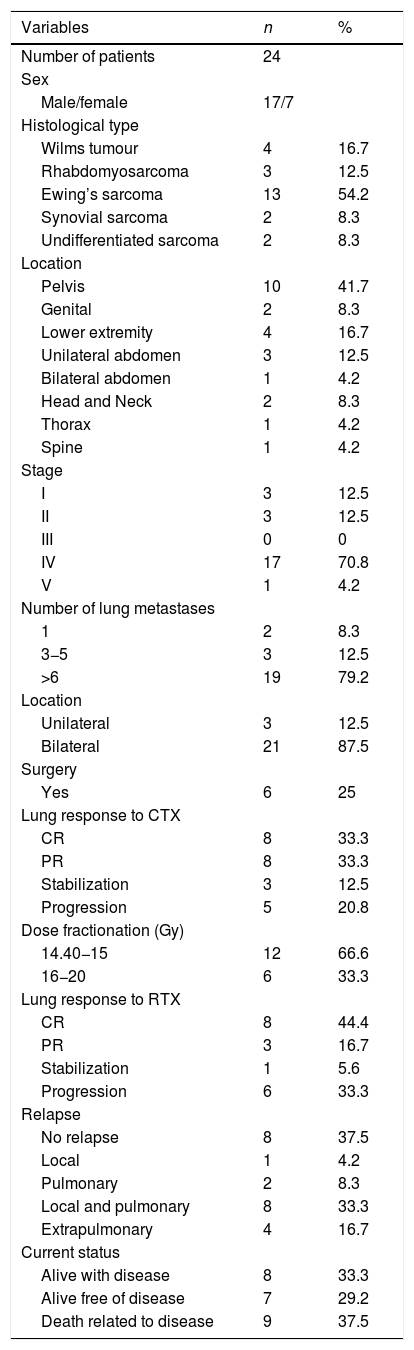
Clinical characteristics of studied patients.
| Variables | n | % |
|---|---|---|
| Number of patients | 24 | |
| Sex | ||
| Male/female | 17/7 | |
| Histological type | ||
| Wilms tumour | 4 | 16.7 |
| Rhabdomyosarcoma | 3 | 12.5 |
| Ewing’s sarcoma | 13 | 54.2 |
| Synovial sarcoma | 2 | 8.3 |
| Undifferentiated sarcoma | 2 | 8.3 |
| Location | ||
| Pelvis | 10 | 41.7 |
| Genital | 2 | 8.3 |
| Lower extremity | 4 | 16.7 |
| Unilateral abdomen | 3 | 12.5 |
| Bilateral abdomen | 1 | 4.2 |
| Head and Neck | 2 | 8.3 |
| Thorax | 1 | 4.2 |
| Spine | 1 | 4.2 |
| Stage | ||
| I | 3 | 12.5 |
| II | 3 | 12.5 |
| III | 0 | 0 |
| IV | 17 | 70.8 |
| V | 1 | 4.2 |
| Number of lung metastases | ||
| 1 | 2 | 8.3 |
| 3−5 | 3 | 12.5 |
| >6 | 19 | 79.2 |
| Location | ||
| Unilateral | 3 | 12.5 |
| Bilateral | 21 | 87.5 |
| Surgery | ||
| Yes | 6 | 25 |
| Lung response to CTX | ||
| CR | 8 | 33.3 |
| PR | 8 | 33.3 |
| Stabilization | 3 | 12.5 |
| Progression | 5 | 20.8 |
| Dose fractionation (Gy) | ||
| 14.40−15 | 12 | 66.6 |
| 16−20 | 6 | 33.3 |
| Lung response to RTX | ||
| CR | 8 | 44.4 |
| PR | 3 | 16.7 |
| Stabilization | 1 | 5.6 |
| Progression | 6 | 33.3 |
| Relapse | ||
| No relapse | 8 | 37.5 |
| Local | 1 | 4.2 |
| Pulmonary | 2 | 8.3 |
| Local and pulmonary | 8 | 33.3 |
| Extrapulmonary | 4 | 16.7 |
| Current status | ||
| Alive with disease | 8 | 33.3 |
| Alive free of disease | 7 | 29.2 |
| Death related to disease | 9 | 37.5 |
CR, complete response; CTX, chemotherapy; PR, partial response; RTX, radiotherapy.
Current indications in clinical trials of WLI.
| Protocol | Wli indication |
|---|---|
| EUROEWING 2012 |
|
| UMBRELLA SIOP-RTSG 2016 |
|
| EpSSG RMS 2005 |
|
Of the 24 children under study, 18 had lung metastases at diagnosis, and 6 at the time of recurrence; the metastases were bilateral in 21. After chemotherapy (CTX), 8 patients exhibited a complete response (CR), 8 a partial response (PR), and the remaining 8 patients, disease progression or stabilization. Six children underwent surgery to remove the lung metastases. Prophylactic WLI was prescribed to 7 patients. The median dose was 16 Gy (range, 14.4-20 Gy). There was only 1 case of toxicity, corresponding to a patient that developed symptomatic pneumonitis.
Six patients did not receive WLI, of who 5 underwent surgery and 1 did not. Only 8 patients achieved a CR after WLI, 6 in the prophylactic WLI group and 2 in the radical WLI group. One patient exhibited pulmonary disease progression after prophylactic WLI. Of the 11 patients that received radical WLI, 7 exhibited pulmonary disease progression during the follow-up. With a median follow-up of 39 months, the 3-year overall survival of the total sample was 73% (89% of the prophylactic WLI group, and 63% of the radical WLI group). Factors that appeared to be associated with a poorer prognosis, but which were not statistically significant, were the presence of metastases at diagnosis, absence of a CR after chemotherapy, or not meeting the criteria for indication of lung surgery.
These data, consistent with other retrospective series in the literature, question the use of WLI in patients with lung metastases after CTX that are not eligible for surgery. The impact on local disease control and overall survival after WLI in patients with measurable lung disease is poor and, although the toxicity of radiotherapy (RTX) is low, the clinical benefits of WLI should be considered.
While prophylactic WLI has proven beneficial in terms of pulmonary disease-free survival (DFS), its influence in OS has not been studied so long. In a study in patients with inoperable metastases of nephroblastoma, Verschuur et al. found that the 5-years OS in the inoperable group was lower compared to survival in patients that underwent resection and received CTX (48% vs 92%; P < .001).1 Dix et al. studied children with nephroblastoma and lung metastases treated with RTX, comparing children with a complete response after CTX that did not receive WLI and children with an intermediate response to CTX that received WLI. In the complete response group, the 4-year DFS and 4-years OS were 79.5% and 96.1%, respectively. In the intermediate response group, the 4-year DFS and OS were 88.5% and 95.4%, respectively.2
Roderberg et al. assessed the benefits of adding WLI to the management of children with lung metastases of rhabdomyosarcoma, and found that WLI was associated with a decrease in pulmonary recurrence (P = .04), but had no impact in OS (WLI, 47% vs no WLI, 30%).3 In children with lung metastases of Ewing Sarcoma treated with RTX, Casey et al. described an improvement in the incidence of pulmonary recurrence, DFS and OS in patients treated with WLI and exclusive lung metastases compared to children with extrapulmonary disease, with a survival free of pulmonary disease of 45%.4 Scobioala et al. analysed the role of WLI in children who had pulmonary recurrence and were rescued with surgery and/or CTX followed by WLI, describing a benefit in the 3-year-OS in patients that exhibited a CR compared to a partial response or stabilisation of the disease (65% vs. 53%; P = .03), an association that was not maintained in the 3-year DFS (54% vs. 49%; P = .10).5
Ongoing clinical trials in solid paediatric tumours (EuroEwing, Umbrella, EpSSG RMS 2005…) continue to include the indication for WLI, despite a lack of evidence of it having a positive impact on OS. Moreover, trial protocols do not adjust the dose of RTX prescribed for macroscopic disease or in case of lack of response to CTX. The only factor that is considered for adjustment of the RTX dose is age (15 Gy vs 18 Gy).
To date, no prospective studies have been published that attempt to clarify the indication of WLI in these patients. Aspects to be considered in the design of future clinical trials include performing surgery or delivering higher RTX doses in measurable metastases with stereotactic radiation therapy techniques and comparing outcomes in disease control and survival, with and without the use of WLI, in patients that are candidates for prophylaxis.
Please cite this article as: Gutiérrez FD, Segundo CGS, Ferreras PS, Catalán MA, Domínguez DR. Irradiación pulmonar total en los tumores sólidos pediátricos: ¿una indicación que ha de revisarse? An Pediatr (Barc). 2021;95:473–475.