Assessment of postoperative pain is a fundamental aspect of post-surgical care. In day surgery paediatric patients, parents are mainly responsible for assessing paediatric pain; however, their estimation may not always be correct, and instruments are needed to help them make a reliable evaluation. The Parent's Postoperative Pain Measurement (PPPM) is a behavioural measure of postoperative pain developed to assist parents with pain assessment. The purpose of this study was to translate the PPPM into Spanish and validate the psychometric properties of the new Spanish version.
MethodA total of 111 paediatric surgery patients aged between 2 and 12 years and 1 parent of each study subject were included. After surgery, each child's level of pain was assessed, and each parent completed the Spanish PPPM scale.
ResultsThe PPPM items in Spanish showed good internal consistency (Cronbach's alpha between 0.784 and 0.900) and scores were closely related to the global pain assessment (Spearman's rho correlation between 0.626 and 0.431). The score decreased from the day of surgery to the first postoperative day, and the scale was successful in discriminating between children who had undergone mild, moderate and severe pain surgeries.
ConclusionsWe conclude that the Spanish version of the PPPM scale evaluated in this study is a suitable tool for assessing paediatric postoperative pain in the home.
La evaluación del dolor postoperatorio es un aspecto fundamental de los cuidados posquirúrgicos. Cuando la cirugía es ambulatoria, los principales responsables de la evaluación del dolor son los padres, pero los padres no siempre pueden evaluar correctamente el dolor de los niños. Esto hace necesario disponer de instrumentos que les ayuden a valorar el dolor postoperatorio de manera fiable. La Parent's Postoperative Pain Measurement (PPPM) es una medida conductual de dolor postoperatorio desarrollada para ayudar a los padres a evaluar el dolor postoperatorio de los niños. El propósito de este trabajo fue traducir al español esta escala y validar las propiedades psicométricas de esta versión en español.
MétodoParticiparon 111 niños de entre 2 y 12 años sometidos a cirugía y uno de sus padres. Tras la operación, se valoró el nivel de dolor de los niños y los padres completaron la escala PPPM en español.
ResultadosLos ítems de la escala PPPM en español mostraron buena consistencia interna (alfa de Cronbach entre 0,784 y 0,900) y las puntuaciones en la escala estuvieron fuertemente relacionadas con la evaluación global del dolor (correlación Rho de Spearman entre 0,626 y 0,431). La puntuación en la escala disminuyó entre el día de la operación y el día siguiente, y discriminó bien entre los niños sometidos a cirugías calificadas como de dolor bajo/moderado y de alto dolor.
ConclusionesConcluimos que la versión española de la escala PPPM que se incluye en este trabajo tiene buenas propiedades psicométricas para evaluar el dolor postoperatorio en el domicilio por parte de los padres.
Changes in the organisation of the public health service have significantly increased the number of procedures carried out in day surgery units. Around 60% to 70% of paediatric pathologies require short, low-risk procedures with minimum blood loss. This, together with a low rate of comorbidities, has made outpatient surgery an attractive option in children. Day surgery has several advantages: it shortens hospital stays, minimises disruptions in the family's routine and benefits the patient's psychosocial well-being. Moreover, the parents of day surgery patients tend to be less anxious and more cooperative.1 Aside from reducing incidence of nosocomial infection, day surgery is cost-effective, helps shorten waiting lists, and has been associated with speedier somatic and psychological recovery times.1 Cost savings, however, should never be made at the expense of quality of care, outcomes, and patient satisfaction.2 In paediatric day surgery, patients are discharged from hospital within a few hours; therefore, postoperative pain management is largely the responsibility of parents. A number of studies have shown that parental pain assessment is not always reliable, and many parents underestimate their child's level of pain3, a situation that could be related with a tendency to under-medicate their children during the postoperative period.4 For this reason, parents need tools that can help them make an accurate assessment.
The Parent's Postoperative Pain Measurement (PPPM) is a postoperative pain behavioural measure developed to help parents in the assessment of postoperative pain in children.5 The PPPM was initially designed to be used with children between 7 and 12 years,6 but subsequent studies5 confirmed the reliability and validity of the scale as a measure of postoperative pain in children from 2 to 12 years. Several studies have confirmed the reliability and validity of the original PPPM;5,7,8 it has been incorporated into clinical practices guidelines,9 and is one of the recommended observational measures for assessing postoperative pain in children and adolescents in the home.10 The PPPM scale has been used in studies for various purposes: assessing differences between surgical techniques8,11; measuring chronic pain in children12,13; examining children's and parent's perspectives in coping with postoperative pain14; examining the relationship between postoperative pain and parents’ perception of their adherence to pain management instructions given at discharge15; understanding parents’ perception of their child's pain16 or the use of non-pharmacological methods to alleviate their child's postoperative pain in the home17; comparing the effect of different anaesthetics18,19; studying the psychological predictors of postoperative sleep patterns in children following day surgery20, or the prevalence and predictors of sleep disturbances following day surgery21; studying the relationship between preoperative anxiety, postoperative pain and other problems22; comparing the parental global impression of acute pain in children against the PPPM score and self-reported pain23; and examining parental pharmacological and non-pharmacological pain management following fracture injuries in children.24 The impact of the PPPM on pain management strategies adopted by the parents of children aged between 1 and 2 years has also been studied.25 Recently, the PPPM was used to assess the prevalence, severity and duration of postoperative pain in children undergoing day surgery.26,27 It has been compared to other pain scales such as the FPS-R,28 and its effectiveness has made it one of the most widely used pain assessment tools in paediatric patients.29
The PPPM has been validated in several languages30,31 but not in Spanish. The aim of this study has been to translate, adapt and validate the psychometric properties of the Spanish version of the PPPM for a sample of children aged between 2 and 12 years.
MethodParticipantsThe study was carried out at the Sant Joan de Dios tertiary teaching hospital in Barcelona, Spain. Children between 2 and 12 years who underwent minor surgery in 2 of the surgical units of the hospital during the data collection period (February–October 2013) and whose parents signed the informed consent form were considered eligible for this study. Parents with insufficient command of Spanish were excluded. In total, 168 children were considered eligible, of whom 111 children were finally included (71 boys and 36 girls, or 64% of eligible subjects), and 1 parent or carer per child (78 mothers [70.3%], 23 fathers [20.7%], and 1 carer [0.9%]). The children were aged between 2 and 12 years (mean age±standard deviation 5.9±2.8 years). All the children had undergone minor surgery and had been discharged from hospital on the day of surgery. The distribution of surgery by type is as follows: abdominal 11 (10%), dental 5 (5%), dermatological 7 (6%), ophthalmic 4 (4%), oncological 5 (5%), ear nose and throat 18 (16%), rheumatological 11 (10%), orthopaedic 11 (10%), urological 29 (26%), others 6 (6%), together with 4 unspecified procedures (4%). An anaesthetist estimated the pain that could be expected from each procedure: 7 (6%) children underwent high-pain surgery, 49 (44%) underwent moderate-pain procedures, and 51 (46%) surgeries were associated with little or not postoperative pain.
MeasurementsParents’ Postoperative Pain Measure (PPPM)The parents were asked to complete the 15 behavioural items of the PPPM, which had been previously translated and adapted to Spanish following the guidelines of the International Test Commission32–34, to ensure it was linguistically, semantically and conceptually equivalent to the English version. The PPPM was translated in 3 phases: (a) 3 translators translated the PPPM into Spanish independently; (b) a summary of the 3 translations was made by consensus to obtain the first draft of the Spanish version of the scale; (c) this version was back-translated into English by a translator not involved in the first stage of translation; (d) a group of healthcare professionals with experience in paediatric pain assessment and knowledge of English discussed the different versions and the back-translation and re-formulated any items that seemed ambiguous or repetitive in Spanish; (e) the 15 items and the instructions in Spanish were read by a group of 10 parents to ensure they were easily understood and correctly interpreted; and (f) the research team reviewed the suggestions and contributions of the parents and healthcare professionals and decided on the final formulation of the items in the Spanish scale, following which the final version was reviewed by 2 Spanish specialists. Table 1 shows the original English version (PPPM) and the Spanish version of the scale (PPPM-ES). This scale and the instructions in Spanish are available at http://gredos.usal.es/jspui/bitstream/10366/123143/1/PPPM-S.pdf.
Original version in English and version in Spanish (PPPM-ES) of the items in the PPPM scale.
| Original English version | Spanish version |
|---|---|
| 1. Whine or complain more than usual? | 1. ¿Lloriquea o se queja más de lo normal? |
| 2. Cry more easily than usual? | 2. ¿Llora más fácilmente de lo normal? |
| 3. Play less than usual? | 3. ¿Juega menos de lo normal? |
| 4. Not do the things he/she normally does? | 4. ¿No hace las cosas que habitualmente hace? |
| 5. Act more worried than usual? | 5. ¿Está más preocupado/a de lo normal? |
| 6. Act more quiet than usual? | 6. ¿Está más callado/a de lo normal? |
| 7. Have less energy than usual? | 7. ¿Tiene menos energía de lo habitual? |
| 8. Refuse to eat? | 8. ¿Se niega a comer? |
| 9. Eat less than usual? | 9. ¿Come menos de lo habitual? |
| 10. Hold the sore part of his/her body? | 10. ¿Sujeta la parte dolorida de su cuerpo? |
| 11. Try not to bump the sore part of his/her body? | 11. ¿Procura no golpear la parte dolorida de su cuerpo? |
| 12. Groan or moan more than usual? | 12. ¿Gime o se lamenta más de lo normal? |
| 13. Look more flushed than usual? | 13. ¿Parece más ruborizado/a de lo normal? |
| 14. Want to be close to you more than usual? | 14. ¿Quiere estar cerca de usted más de lo normal? |
| 15. Take medication when he/she normally refuses? | 15. ¿Toma la medicación cuando normalmente la rechaza? |
Pain levels were assessed on the basis of scales used in the hospital to evaluate pain by age group: for children from 2 to 4 years, parents were asked to assess pain on a scale of 0 (no pain) to 10 (worse pain imaginable). For the 4–6 age group, parents were asked to assess their child's pain on the same 0–10 scale, while the children reported their own pain level on the revised FACES pain scale.35 On this scale, parents asked their child to choose one of 6 faces (face 1=no pain, face 6=worst possible pain) to indicate how they felt that day. The 6 faces scale was also used in children between 6 and 8 years, or in the case of non-cooperative adults. Children between 8 and 12 years were asked to assess their pain on the 0–10 scale. The scores obtained from the 6 faces scale were adapted to those obtained from the 0–10 scale in order to compare the pain level assessment of the different age groups.
ProcedureAfter surgery, once the child had been made comfortable and the parents were no longer concerned for the child's welfare, a nurse explained the study to the parents and invited them to take part. If they agreed, they were asked to read and sign an informed consent form before they returned home with their child. Children over the age of 7 were also asked to sign an informed consent form, but in this case they were allowed to sign it at home if they were unable to do so in the hospital. Parents and children were taught how to use the pain scales and complete the questionnaire (in the case of children, they were told to simply express how they felt). Parents were given copies of the PPPM-ES questionnaire and pain scale, together with a stamped addressed envelope to post the completed forms back to the hospital. They were asked to complete 2 questionnaires: the first on the day of surgery (once the child was at home), and the second on the following day. Both questionnaires assessed the child's behaviour and reactions during the day, and therefore had to be completed in the evening, once the child was in bed for the night. The day after surgery, during the routine phone call to enquire after the health of the child, parents were asked whether they had completed the forms, and were reminded to complete them again that evening, and then post the questionnaires and pain scales back to the hospital. The study was approved by the hospital's Independent Ethics Committee.
Statistical analysisA frequency analysis was performed on each of the 15 items in the PPPM-ES. Eta correlation coefficients were used (for relationships between nominal and ordinal variables) to examine the relationship between each item in the PPPM-ES and the global assessment of pain. Cronbach's alpha coefficient was used to examine the internal reliability of the measurement in both (younger and older) age groups. T tests for dependent samples were used to examine the differences in PPPM-ES scores and pain scores between the first day of surgery and the following day, and also differences in PPPM-ES scores when analysed by age and type of surgery.
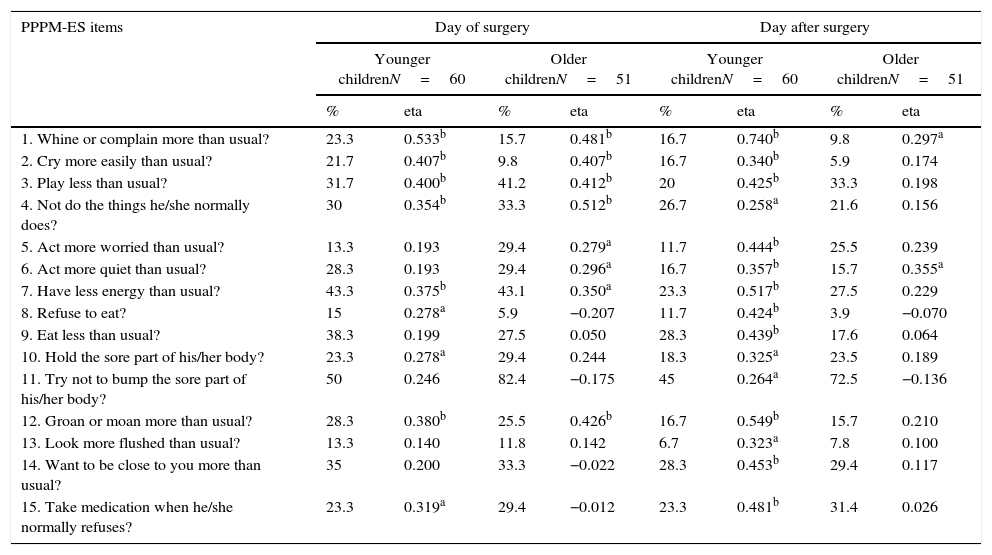
ResultsAnalysis of itemsTable 2 shows the scores of each item in the PPPM-ES questionnaire, the percentage of affirmative answers, and the correlation between each item and the pain scale score obtained from the 2 younger groups (2–6 years) and the older children (7–12 years) on the day of surgery and the following day.
Frequency of each item and correlation between each item and pain assessment on the day of surgery and the following day for each group of children.
| PPPM-ES items | Day of surgery | Day after surgery | ||||||
|---|---|---|---|---|---|---|---|---|
| Younger childrenN=60 | Older childrenN=51 | Younger childrenN=60 | Older childrenN=51 | |||||
| % | eta | % | eta | % | eta | % | eta | |
| 1. Whine or complain more than usual? | 23.3 | 0.533b | 15.7 | 0.481b | 16.7 | 0.740b | 9.8 | 0.297a |
| 2. Cry more easily than usual? | 21.7 | 0.407b | 9.8 | 0.407b | 16.7 | 0.340b | 5.9 | 0.174 |
| 3. Play less than usual? | 31.7 | 0.400b | 41.2 | 0.412b | 20 | 0.425b | 33.3 | 0.198 |
| 4. Not do the things he/she normally does? | 30 | 0.354b | 33.3 | 0.512b | 26.7 | 0.258a | 21.6 | 0.156 |
| 5. Act more worried than usual? | 13.3 | 0.193 | 29.4 | 0.279a | 11.7 | 0.444b | 25.5 | 0.239 |
| 6. Act more quiet than usual? | 28.3 | 0.193 | 29.4 | 0.296a | 16.7 | 0.357b | 15.7 | 0.355a |
| 7. Have less energy than usual? | 43.3 | 0.375b | 43.1 | 0.350a | 23.3 | 0.517b | 27.5 | 0.229 |
| 8. Refuse to eat? | 15 | 0.278a | 5.9 | −0.207 | 11.7 | 0.424b | 3.9 | −0.070 |
| 9. Eat less than usual? | 38.3 | 0.199 | 27.5 | 0.050 | 28.3 | 0.439b | 17.6 | 0.064 |
| 10. Hold the sore part of his/her body? | 23.3 | 0.278a | 29.4 | 0.244 | 18.3 | 0.325a | 23.5 | 0.189 |
| 11. Try not to bump the sore part of his/her body? | 50 | 0.246 | 82.4 | −0.175 | 45 | 0.264a | 72.5 | −0.136 |
| 12. Groan or moan more than usual? | 28.3 | 0.380b | 25.5 | 0.426b | 16.7 | 0.549b | 15.7 | 0.210 |
| 13. Look more flushed than usual? | 13.3 | 0.140 | 11.8 | 0.142 | 6.7 | 0.323a | 7.8 | 0.100 |
| 14. Want to be close to you more than usual? | 35 | 0.200 | 33.3 | −0.022 | 28.3 | 0.453b | 29.4 | 0.117 |
| 15. Take medication when he/she normally refuses? | 23.3 | 0.319a | 29.4 | −0.012 | 23.3 | 0.481b | 31.4 | 0.026 |
The results of the Cronbach's alpha test in the younger sample varied between 0.842 and 0.899 for the day of surgery and the following day, respectively, and between 0.784 and 0.853 for the day of surgery and the following day in the older group. The results indicate that the scale is reliable.
ValidityCorrelation between global pain assessments and the PPPM-ES scoresSpearman's Rho correlation coefficients between PPPM-ES scores and pain scales for younger children were 0.519 and 0.666 for the day of surgery and the following day, respectively (p<0.01) and 0.501 (p<0.01) for the day of surgery and 0.297 (p<0.05) for the following day in the older group.
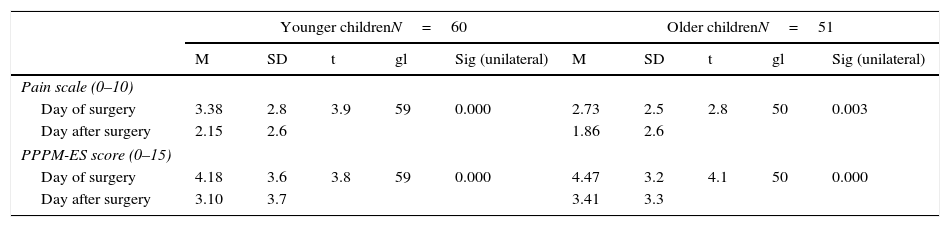
Differences between mean pain scale and PPPM-ES scoresIn Table 3, the means±standard deviations of the pain scale (0–10) score and PPPM-ES score (0–15) are shown for each age group (younger and older children) and each evaluation time (day of surgery and first day post-surgery).
Mean and standard deviation of pain scale and PPPM-ES score in the 2 age groups at the 2 assessment times.
| Younger childrenN=60 | Older childrenN=51 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| M | SD | t | gl | Sig (unilateral) | M | SD | t | gl | Sig (unilateral) | |
| Pain scale (0–10) | ||||||||||
| Day of surgery | 3.38 | 2.8 | 3.9 | 59 | 0.000 | 2.73 | 2.5 | 2.8 | 50 | 0.003 |
| Day after surgery | 2.15 | 2.6 | 1.86 | 2.6 | ||||||
| PPPM-ES score (0–15) | ||||||||||
| Day of surgery | 4.18 | 3.6 | 3.8 | 59 | 0.000 | 4.47 | 3.2 | 4.1 | 50 | 0.000 |
| Day after surgery | 3.10 | 3.7 | 3.41 | 3.3 | ||||||
When comparing the 2 assessment times, the significance test shows that scores are significantly lower the day after surgery on both the pain scale and PPPM-ES scales for both age groups (Table 3).
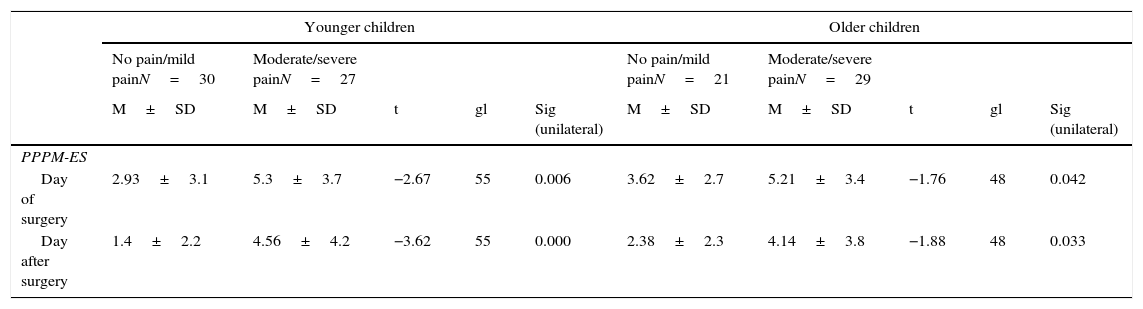
Table 4 shows the means±standard deviation of PPPM-ES scores on the day of surgery (day 0) and the first postoperative day (day 1) by age group (younger and older children) and type of surgery (no/mild pain or moderate/severe pain). The results show significant differences between PPPM-ES scores for no/mild pain and moderate/severe pain procedures in both younger and older children. (Table 4).
Mean±standard deviation of PPPM-ES scores on the day of surgery and first postoperative day by type of surgery and age group.
| Younger children | Older children | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| No pain/mild painN=30 | Moderate/severe painN=27 | No pain/mild painN=21 | Moderate/severe painN=29 | |||||||
| M±SD | M±SD | t | gl | Sig (unilateral) | M±SD | M±SD | t | gl | Sig (unilateral) | |
| PPPM-ES | ||||||||||
| Day of surgery | 2.93±3.1 | 5.3±3.7 | −2.67 | 55 | 0.006 | 3.62±2.7 | 5.21±3.4 | −1.76 | 48 | 0.042 |
| Day after surgery | 1.4±2.2 | 4.56±4.2 | −3.62 | 55 | 0.000 | 2.38±2.3 | 4.14±3.8 | −1.88 | 48 | 0.033 |
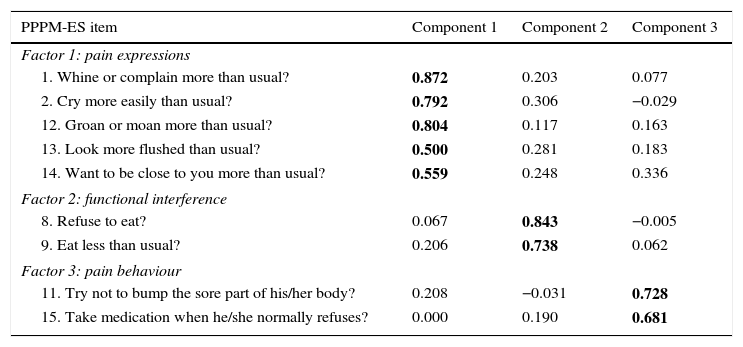
A principal components factor analysis was performed on the 15-item parent questionnaire, which yielded 3 components with eigenvalues greater than 1. The first of these explained 41.7% of the variance, the second 8.5%, and the third 7.5% (all before rotation). The first component (pain expressions) comprises 5 items, and the second and third components (functional interference and pain behaviour) comprise 2 items each (Table 5).
Factorial analysis: extraction of the main components of the PPPM-ES.
| PPPM-ES item | Component 1 | Component 2 | Component 3 |
|---|---|---|---|
| Factor 1: pain expressions | |||
| 1. Whine or complain more than usual? | 0.872 | 0.203 | 0.077 |
| 2. Cry more easily than usual? | 0.792 | 0.306 | −0.029 |
| 12. Groan or moan more than usual? | 0.804 | 0.117 | 0.163 |
| 13. Look more flushed than usual? | 0.500 | 0.281 | 0.183 |
| 14. Want to be close to you more than usual? | 0.559 | 0.248 | 0.336 |
| Factor 2: functional interference | |||
| 8. Refuse to eat? | 0.067 | 0.843 | −0.005 |
| 9. Eat less than usual? | 0.206 | 0.738 | 0.062 |
| Factor 3: pain behaviour | |||
| 11. Try not to bump the sore part of his/her body? | 0.208 | −0.031 | 0.728 |
| 15. Take medication when he/she normally refuses? | 0.000 | 0.190 | 0.681 |
Bold values indicate significant correlation level 0.01.
The aim of this study was to test the reliability and validity of the Spanish version of the PPPM scale in a group of paediatric patients aged between 2 and 12 years undergoing minor surgery. The correlation analysis shows that in most items the PPPM-ES significantly correlated with levels of pain reported by parents in both age groups. These results were similar to those obtained with the original English version of the tool. The Cronbach's alpha score for reliability was positive, showing good internal consistency for both assessment times (day of surgery and first postoperative day) and groups (younger and older children). The PPPM-ES was also shown to be valid, with a significant decrease in pain scores in both age groups from the day of surgery to the first postoperative day. The means of PPPM-ES scores also differed significantly between children undergoing moderate/severe pain surgery and those receiving no/mild pain procedures. The factorial analysis showed that the 15-item questionnaire comprises 3 principal components.
This study has several limitations. First, no children with developmental difficulties were included, which prevented us from determining the validity and reliability of the scale in this population. Further studies are needed to explore parental postoperative pain assessment in developmentally handicapped children. Secondly, children undergoing repetitive or complex surgical procedures were not included. Pain management is particularly important in this patient group, and further research is needed to test the psychometric properties of the scale in this context. Finally, in contrast to the original English scale, PPPM-ES scores before and after administration of analgesics were not compared7 The conditions of work did not permit this, and this limitation should be addressed in future studies. Despite these limitations, we believe that this study has shown that the Spanish version of the PPPM-ES is a valid and reliable tool for assessing paediatric postoperative pain.
FundingThis study has received funding from the Health Council of Castilla and León Regional Board (Consejería de Sanidad de la Junta de Castilla y León) (ref. GES/SA03/13).
Conflict of interestThe authors declare that there are no conflicts of interest.
Please cite this article as: Ullan AM, Perelló M, Jerez C, Gómez E, Planas MJ, Serrallonga N. Validación de la versión española de la escala de evaluación del dolor postoperatorio Parent's Postoperative Pain Managament. An Pediatr (Barc). 2016;84:106–113.